Review Article
Legislative control of Quinoa in the United Kingdom and European Union
School of Applied Sciences, London South Bank University, Borough Road, London SE1 0AA, United Kingdom
*Corresponding author: Delia Ojinnaka, School of Applied Sciences, London South Bank University, Borough Road, London SE1 0AA, United Kingdom, Tel: 020-7815-6255, Fax: 020-7815-7999, E-mail: ojinnad@lsbu.ac.uk
Received: September 26, 2016 Accepted: October 4, 2016 Published: February 27, 2017
Citation: Ojinnaka D. Legislative control of Quinoa in the United Kingdom and European Union. Madridge J Food Technol. 2017; 2(1): 52-56. doi: 10.18689/mjft-1000108
Copyright: © 2017 The Author(s). This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Food safety control in relation to quinoa (Chenopodium quinoa Willd) as food and food ingredient was discussed and evaluated through an examination of the current statutory provisions at United Kingdom (UK) and European Union (EU) levels. The risk, if any, posed by quinoa grains, from production to consumption was identified and suggestions for compliance were made. Quinoa though novel in the UK and marketed loosely as super food, apparently does not warrant any specific (vertical) food safety control requirements and thus is not restricted by the Novel Food Regulation (EC) No. 258/97. This assertion is supported through the composition; the constituents are non-toxic, consumption of quinoa in South America dates back to several centuries and there is no reported adverse food safety incident. However, quinoa, must meet the food safety requirements as stated in the Food Safety Act 1990 and/or Regulation (EC) 178 /2002 on general food law.
Keywords: Quinoa; EU food safety legislation; The Food Safety Act 1990; Regulation (EC) 178 /2002; Regulation (EC) 258/97.
Introduction
Quinoa (Chenopodium quinoa Willd) is an annual crop that originates from the Andes, South America, covering Argentina, Bolivia, Chile, Colombia, Ecuador, Peru and Venezuela and with Peru and Bolivia having the most germplasm accession. It is currently cultivated in Spain [1] and Malawi [2] where the potential contribution of quinoa to food security and sustainability was explored. Quinoa is among the crop selected for food security consideration by the Food and Agriculture Organisation (FAO) [3] as the protein quality of the seed is deemed to be better than those of cereals. Quinoa contains more lysine and methionine than cereals [4] and furthermore, can thrive in adverse agroecological conditions. Cultivation tests have been carried out with some success across the world [5-7].
Quinoa grain has been used as food over several centuries; the grain is consumed as a staple and is widely used as an ingredient in the production of ‘chicha’ beer [8], breakfast foods and soups [9]. It may be used as an alternative to rice, beans, wheat and lentils. The use of quinoa in the production of gluten-free flour confectionery is being explored.
Quinoa seed is making a gradual appearance in the United Kingdom (UK) food market, and is sold as a super food / super grain under the umbrella of healthy and vegetarian food and consequently commanding a premium price. The average cost being about 80 pence per 100 grams, thus 20% more than a regular basmati rice and much more than a long grain rice. At the moment it is a niche market and is sold on its own; white, red or mixed and also in combination with other grains; rice, wheat, couscous and in the production of vegetarian rolls and chips. Thus the range of the products is wide and endless. The versatility is attributable to the subtle aroma and acceptable texture. Quinoa seems to present a real alternative to conventional cereals such as wheat, oat, maize and rice as well as traditional foods such as couscous and pasta. It is readily adapted as a food ingredient in the United States and Europe [10].
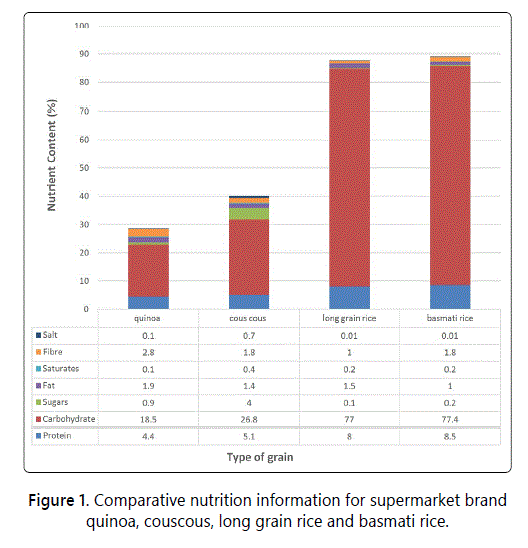
The nutritional composition of quinoa is equally comparable (Figure 1) with those of rice and couscous with quinoa being significantly lower in total carbohydrate and higher in fibre. The values were obtained from the nutrition information on the label of products sold in a UK supermarket and are based on the AOAC standard methods [11]. The values of non-nutrients and water were not stated on the labels. It was not possible to use mean values as the supermarket did not stock a range of quinoa grain.
EU food safety control and requirements
The safety of evaluation of quinoa as food and food ingredient indicated that special control is unnecessary. However, quinoa seeds and products are liable to the general food safety control. Food safety is one of the key issues of food control, from the consumers, producers and food law regulators points of view. Success lies in the introduction and implementation of effective statutory and non-statutory systems of control, ranging from microbiological, chemical and physical safety. Modern food control is based on the precautionary principle, the scope ranging from farm to plate, including all stages of production, processing, delivery and the sale of foods. The principles of control based on prevention through self-regulation rather than detection and treatment. Quinoa is no exception to these measures.
Control exists at various levels; national, European Union (EU) and global through the Codex Alimentarius Commission. In the UK control rests with the Food Standards Agency (FSA), Department of Health (DoH) and Department of Environment, Food and Rural Affairs (DEFRA). Similar function is performed at the EU level by the European Food Safety Authority (EFSA).
The safety and wholesomeness of food are of paramount importance to every consumer. There are several links between diet and longevity, ailment and wellness [12-18]. Specific foods, ingredients and the chemicals they contain can have a direct impact on health. The responsibility of assuring the safety of food lies primarily with government and the industry. In todayʼs food industry, all aspects of the production, storage and distribution of food must be effectively controlled, not only to assure safety and wholesomeness, but also to ensure efficient and consistent manufacture, at the lowest possible cost. In the meantime, consumers, in particular those of the developed nations are demanding a greater assurance of safety as well as more information on which to base their choice.
Overall, national and international legislation provides the basis for much of this control. The legislation is an essential element for an effective food safety and control system.
Statutory control, irrespective of the source deals specifically with three main areas; public health, standards and labelling. Thus all the food produced for sale must be safe and not injurious to health by meeting the food safety requirements as stated in section 7 of the Food Safety Act 1990 (FSA) as amended (Legislation.gov.uk) [19] and article 14 of Regulation (EC) 278 / 2002 on the general principles and requirements of food law and procedures in matters of food safety (Europa. Eu.int/ eur-lex/en/archive/2002) [20]. This Regulation has a direct application in all Member States.
The Food Safety Act 1990 is a primary Act which provides the framework for all food legislation in Great Britain with the objective of ensuring that all food produced for sale is safe to eat and not misleadingly presented or labelled. The requirements are expressly stated in section 7 which provides an offence of rendering foods injurious to health through; (a) addiction of any article or substance to the food (b) use of any article or substance as an ingredient in the preparation of the food, (c) abstraction of any constituent from the food and (d) subjecting the food to any other process or treatment. Injury means any impairment to health whether permanent or temporary and in its determination, probable and cumulative effects on the consumers are considered.
The FSA 1990 has been amended to bring it in line with Regulation (EC) 178/2002 in which similar provisions are made in articles 14 and 16. Article 14 states that, ‘food shall not be placed on the market if it is unsafe and food shall be deemed to be unsafe if it is considered to be injurious to health and unfit for human consumption’. Article 16 states that ‘without prejudice to more specific provisions of food law, the labelling, advertising and presentation of food or feed, including their shape, appearance or packaging, the packaging materials used, the manner in which they are arranged and the setting in which they are displayed, and the information which is made available about them through whatever medium, shall not mislead consumers’.
Thus, a legal responsibility is imposed on food business operators to meet the food safety requirements.
The general principle in a nutshell is that, there is no 100% safety and 0% risk. Food businesses must have an effective food safety management system in place at all stages. The systems are generally based on hazard analysis critical control points (HACCP) and thus provides a means of self-regulation. These provisions ensure a high degree of protection of public health and protection against fraud.
The commercial quinoa appears to be intrinsically compliant, though there is a concern about saponin which is found in the seed coats and can lead to stomach irritation [21]. The value of saponin ranges from 0.9% to 1.4% in bitter varieties [21,22]. Saponin is a toxic glycoside with bitter taste; the presence of such toxicant in foods may render the food injurious to health and thus breach the legal provision with consequent penalties. This is only speculative because in the UK and EU, there is no legislative control for saponin, no maximum residue level is recommended. The toxicity and bitterness of the grains can be minimised by processing and washing with water. Saponin is water soluble and concentrated on the skin. In fact, Gómez-Caravaca et al. [23] showed that sweet quinoa with a saponin content of less than 0.11% can be obtained by process pearling at an abrasion rate of 30%. Thus the adverse effect of saponin can be minimised by washing or rinsing with water and removing the seed coat.
EU food standards and labelling requirements
There are minimum legal standards, which are set to control food adulteration and ensure authenticity. Food adulteration renders the food non-authentic and may in some instances, present no hazard to health but involves the consumers being sold foods that are below the minimum standard. Typical examples are watering down of fruit juices, inaccurate or overdeclaration of the meat content of comminuted products such as burgers and sausages and addition of corn syrup to pure honey. The composition and labelling of fruit juices and honey are regulated by vertical legislation namely; Council Directive 2001/112/EC relating to fruit juices and certain similar products intended for human consumption and Council Directive 2001/110/EC relating to honey, respectively. These EU directives are implemented in England respectively by the Fruit Juices and Fruit Nectars (England) Regulations 2013 and the Honey (England) Regulations 2015 (Legislation.gov.uk) [19]. There is no need for a vertical legislation relating to quinoa and quinoa products as they are less susceptible to adulteration. The consumption rate is increasing but low.
Food labels are required to give the necessary information to enable the consumer to make an informed choice in relation to the purchase or consumption of the food. The regulations controlling labelling ensure that the labels are not misleading and conform to the given standards. Quinoa and quinoa products have to comply with Regulation (EU) 1169/2011 on the provisions of food information to the consumer (http://ec.europa.eu/food/safety/labelling_nutrition/labelling_legislation/index_en.htm) which is implemented in England by the Food Information Regulation 2014. Article 9 and annex II of Regulation (EU) 1169/2011, listed the mandatory food information as shown (Table 1).
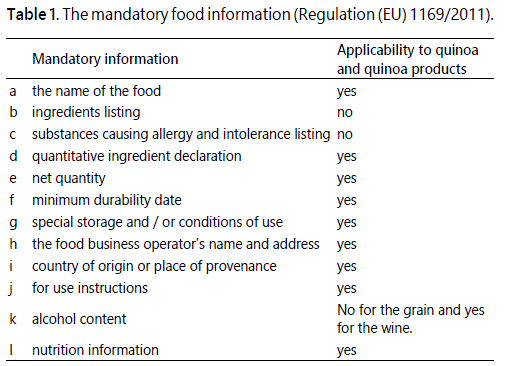
Quinoa as a single ingredient will not be required to comply with the ingredient listing but will comply when used as an ingredient. Annex II listed substances causing allergy or intolerance as; cereals containing gluten (namely, wheat, rye, barley, oats, spelt, kamut or their hybridised strains, crustaceans, fish eggs peanuts soybeans milk nuts (namely, almond, hazelnut, walnut, cashew, pecan nut, brazil nut, pistachio nut, macadamia nut and Queensland nut), celery, mustard, sesame seeds, sulphur dioxide and sulphites at levels above 10 mg/kg or 10 mg/litre expressed as SO2, lupin and molluscs. Quinoa is not included in the food allergen list, however, allergen advice will be necessary if it is treated with sulphites and sulphur dioxide at the levels stipulated above.
The requirement for nutrition information in England comes into force in December 2016. Nutrition labelling, however, remains a mandatory requirement once a claim is made, schedule 7 of the Food Information Regulation 2014 stated that nutrition information should be provided where a nutrition or health claim is made. The nutrition information is supposed to justify the claim (Legislation.gov.uk, 2015). Nutrition labelling is provided by the Council Directive on nutrition labelling for foodstuffs (90/496/EEC) and nutrition and health claims are provided by Regulation (EC) No 1924/2006 of the European Parliament and of the Council on nutrition and health claims made on foods. The former provides for the format of a nutrition label whereas the latter defines nutrition and health claims under article 1 thus; ‘nutrition claim means any claim which states, suggests or implies that a food has particular beneficial nutritional properties due to the energy it provides and/ or nutrient it contain’. Similarly, health claim ‘means any claim that states, suggests or implies that a relationship exists between a food category, a food or one of its constituents and health’ and includes a ‘reduction of disease risk claim’.
In relation to quinoa, possible claims may include; low in energy, low in sugar, high in protein and high in fibre but a closer examination of the composition suggests not, as the amounts of protein and fibre are not exceptional high and furthermore there is no concrete evidence to show that consumption of quinoa and quinoa products uniquely impacts on the physiology and health of the consumer. Any claim on quinoa, alluding to these proprieties will be false. The key requirements for a claim as made in the annex of Regulation (EC) No 1924/2006 of the European Parliament and of the Council on nutrition and health claims made on foods, as may be applied to quinoa is summarised (Table 2).
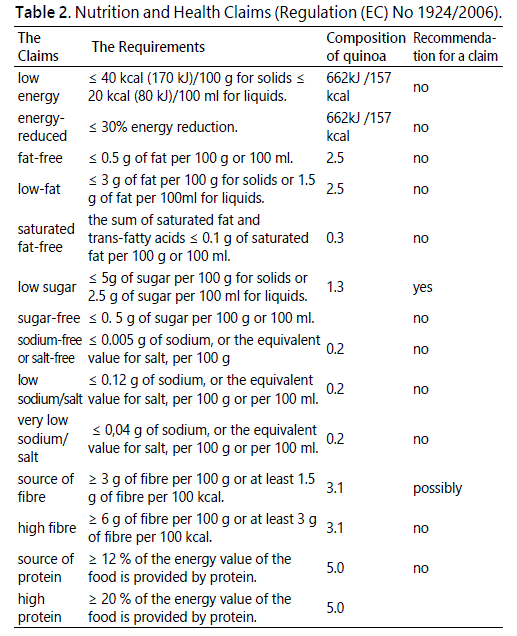
From the table it is apparent that claims for low calories, low fat, low sugar, high protein and high fibre cannot be made as on average the values for quinoa do not meet the legal provisions. However a ‘source of fibre’ and low sugar claims can be made as the fibre content is within the legal limit.
EU contaminants and residues in foods requirements
The presence of contaminants and residues in foods above the maximum residue level (MRL), breaches the provisions of the Food Safety Act 1990 as amended, subsequent regulations, Regulation (EC) 178 /2002 and Commission Regulation (EC) 1881/2006 setting maximum levels for certain contaminants in food stuffs.
Under the FSA 1990, it is an offence to render food injurious to health (S7) and this may occur through the presence of contaminants and residues. In a similar way, Regulation (EC) 178 /2002 states that, ‘food shall not be placed on the market if it is unsafe and shall be deemed to be unsafe if it is considered to be injurious to health and unfit for human consumption’. Furthermore, article 1 the Commission Regulation (EC) 1881/2006 prohibits the placing of foodstuffs in the market if they contain a contaminant listed in the annex at a level exceeding the maximum level set out in the annex.
The levels set for each contaminant or residue is deemed to be safe and presents no toxicological hazard for human health. Safety is further assured by the fact that maximum residue level (MRL) values are usually less than the acceptable daily intake (ADI) and tolerable daily intake (TDI). Foods may contain contaminants and residues which may be carcinogenic, geno toxic and irritant.
A wide range of chemicals are used to improve agricultural yield through pest control, disease control and growth enhancement can be toxic. Human exposure to contaminants and residues is usually through consumption of contaminated foods and the environment. Food and food products are exposed to chemical contaminants through agricultural and non-agricultural industries. Contaminants from the environmental sources may contaminant the soil and water which in turn will contaminant crops and residues may remain in the crops.
Residues are defined as chemicals that are present in food materials as a result of treatment of the plant and animal sources with some chemical compounds. Typical examples are the agrochemicals such as pesticides, fertilisers, growth hormones and therapeutic drugs. Consequently, these residues may be present in the diets of human. Residues may include the parent compounds and their metabolites. The toxicity of the parent compounds may differ from that of their metabolites. Contamination may also occur during production, processing, preparation, treatment, packaging, transport or holding of the foodstuffs.
Quinoa like other grains, nuts and fruits such as maize, groundnut, rice and soya beans is susceptible to fungal growth [24] and thus may contain mycotoxins which cause mycotoxicosis in animals including human [25]. Aflatoxins are of major concern because of their potency, they are very toxic, mutagenic, teratogenic and carcinogenic compounds. Human exposure to aflatoxins is usually through consumption of contaminated foods.
Aflatoxins are one of the major contaminants of food world-wide. Different legislative controls are in place to limit the level of aflatoxins in foods. The general trend is for developed countries to set lower tolerance levels than developing countries. The tolerance levels range from 5 µg kg-1 to 30 µg kg-1. In the EU, under Regulation (EC) 1881/2006, the maximum level for total aflatoxins in cereals and similar products is 4 µg kg-1. Quinoa and quinoa products are susceptible to chemical contaminations at all stages, from farm to plate. Hence these regulatory controls will apply.
Conclusion
This review concludes that commercial quinoa and quinoa products as sold and consumed in the United Kingdom and Europe Union do not require a vertical legislation as long as they comply with the provisions of the Food Safety Act 1990 as amended and Regulation (EC) 178 /2002 on the general principles and requirements of food law and procedures in matters of food safety. Furthermore, the European Commission Novel Food Catalogue stated that, quinoa does not require authorisation under the Novel Food Regulation (EC) 258/97 as it is widely consumed and to a great extent before the legislation came into force on 15th of May 1997. No safety evaluation is required, though assessment of the composition showed that the outer skin contains saponin, a toxic and bitter glycoside. Toxicity and bitterness are reduced by processing [10]. It has been shown that, the level of saponin can be drastically reduced by milling and washing with water. Quinoa is fairly high in fibre, hence a possible claim for ‘high in fibre’ can be made. Similarly a claim for low in sugar and gluten-free can be made. The protein content is similar to that of many cereals; about 16%, thus not warranting a claim in that regard. Compliance with the legislation concerning organic and genetically modified foods is also necessary for organic and genetically modified quinoa.
Quinoa as food and food ingredient posed little risk to public health, though cautioned is advised when taking quinoa with compounds that have antioxidant and fat lowering properties, as quinoa exhibits these properties [26]. Quinoa is freely marketed and consumed in the United Kingdom and the trend is upwards and pushed by the consumersʼ quest for something new, curiosity and eagerness to culinary experimentation.
References
- Maliro MFA, Guwela V. Quinoa Breeding in Africa: History, Goals and Progress. In: Murphy KS, Matanguihan J (eds). Quinoa: Improvement and Sustainable Production. Hoboken, New Jersey: Wiley Blackwell 2015.
- Jacobsen SE. The worldwide potential of quinoa (Chenopodium quinoa Willd). Food Reviews International. 2003; 19: 167-177.
- Fleming JE, Galwey NW. Quinoa (Chenopodium quinoa). In: Williams JT (ed). Cereals and Pseudocereals. Champman & Hall, London. 1995: 383.
- Bhargava A, Shukla S, Ohri D. Genetic variability and interrelationship among various morphological and quality traits in quinoa (Chenopodium quinoa Willd.). Field Crops Res. 2007; 101(1): 104-116. doi: 10.1016/j.fcr.2006.10.001
- Jacobsen SE, Jørgensen I, Stølen O. Cultivation of quinoa (Chenopodium quinoa) under temperate climatic conditions in Denmark. J Agric Sci. 1994; 122: 47-52. doi: 10.1017/S0021859600065783
- Pulvento C, Riccardi M, Lavini A, d'Andria R, Iafelice G, Marconi E. Field trial evaluation of two Chenopodium quinoa genotypes grown under rain-fed conditions in a typical Mediterranean environment in South Italy. J Agron Crop Sci. 2010; 196(6): 407-411. doi: 10.1111/j.1439-037X.2010.00431.x
- Simmonds NW. The grain chenopods of the tropical American highlands. Econ Bot.1965; 19(3): 223-235. doi: 10.1007/BF02914309
- Repo CR, Espinoza C, Jacobsen SE. Nutritionl value and use of the Andean crops quinoa (Chenopodium quinoa) and kañiwa (Chenopodium pallidicaule). Food Rev Int. 2003; 19(1-2): 179-189. doi: 10.1081/FRI-120018884
- Schlick G, Bubenhein DL. Quinoa: Candidate crop for NASA'S controlled ecological life support system. In: Janick J (ed). Progress in new crops. ASHS Press Arlington VA. 1996: 632-640.
- Association of Official Analytical Chemists. Official methods of analysis. Arlington, USA: AOAC 1990.
- Marcus JB. Diet and disease: healthy choices for disease prevention and diet management: practical applications for nutrition, food science and culinary professionals. In: Culinary Nutrition. 2013: 371-430.
- Aruoma OI, Coles LS, Landes B, Repine JE. Functional benefits of ergothioneine and fruit and vegetable-derived nutraceuticals: overview of the supplemental issue contents. Prev Med. 2012; 54: S4-S8. doi: 10.1016/j.ypmed.2012.04.001
- Benford DJ. Risk assessment of chemical contaminants and residues in foods. Persistent Organic Pollutants and Toxic Metals in Foods. 2013: 173-187.
- Clarke R, Connolly L, Frizzell C. Elliott CT. Challenging conventional risk assessment with respect to human exposure to multiple food contaminants in food: A case study using maize. Toxicol Lett. 2015; 238(1): 54-64. doi: 10.1016/j.toxlet.2015.07.006
- Keeney JT, Butterfield DA. Vitamin D deficiency and Alzheimer disease: common links. Neurobiol Dis. 2015; 84: 84-98. doi: 10.1016/j.nbd.2015.06.020
- Maderuelo-Fernandez JA, Recio-Rodríguez JI, Patino-Alonso MC, et al. Effectiveness of interventions applicable to primary health care settings to promote mediterranean diet or healthy eating adherence in adults: a systematic review. Prev Med. 2015; 76: S39-S55. doi: 10.1016/j.ypmed.2014.12.011
- Steele M, Dow L, Baxter G. Promoting public awareness of the links between lifestyle and cancer: a controlled study of the usability of health information leaflets. Int J Med Inform. 2011; 80(12): e214-e229. doi: 10.1016/j.ijmedinf.2011.08.012
- Legislation.gov.uk. UK public general acts. 2015.
- Europa. 2015.
- Ruales J, Nair BM. Saponins, phytic acid, tannins and protease inhibitors in quinoa (Chenopodium quinoa, Willd) seeds. Food Chem. 1993; 48(2): 137-143. doi: 10.1016/0308-8146(93)90048-K
- Gee JM, Price KR, Ridout CL, Wortley GM, Hurrel RF, Johnson IT. Saponins of Quinoa (Chenopodium quinoa): Effects of processing on their abundance in quinoa products and their biological effects on intestinal mucosal tissue. J Sci Food Agric. 1993; 63(2): 201-209. doi: 10.1002/jsfa.2740630206
- Gómez-Caravaca AM, Iafelice G, Verardo V, Marconi E, Caboni MF. Influence of pearling process on phenolic and saponin content in quinoa (Chenopodium quinoa Willd). Food Chem. 2014; 157: 174-178. doi: 10.1016/j.foodchem.2014.02.023
- Pappier U, Fernández Pinto V, Larumbe G, Vaamonde G. Effect of processing for saponin removal on fungal contamination of quinoa seeds (Chenopodium quinoa Willd.). Int J Food Microbiol. 2008; 125(2): 153-157. doi:10.1016/j.ijfoodmicro.2008.03.039
- Schollenberger M, Müller HM, Rüfle M, Suchy S, Planck S, Drochner W. Survey of Fusarium toxins in foodstuffs of plant origin marketed in Germany. Int J Food Microbiol. 2005; 97: 317-326.
- Dini I, Tenore GC, Dini A. Antioxidant compound contents and antioxidant activity before and after cooking in sweet and bitter Chenopodium quinoa seeds. LWT- Food Science and Technology. 2010; 43(3): 447-451. doi: 10.1016/j.lwt.2009.09.010


