Case Report
Cutaneous Metastasis from an Asymptomatic Breast Cancer Masquerading as an Infected Sebaceous Cyst
Department of Pathology, Kalpana Chawla Government Medical College, Karnal, Haryana, India
*Corresponding author: Sonam Sharma, Assistant Professor, Department of Pathology, Kalpana Chawla Government Medical College, Karnal, Haryana, India, Tel: +919999841393, E-mail: drsonamsharma@gmail.com
Received: January 19, 2018 Accepted: February 3, 2018 Published: February 9, 2018
Citation: Sharma S, Sonali. Cutaneous Metastasis from an Asymptomatic Breast Cancer Masquerading as an Infected Sebaceous Cyst. Madridge J Dermatol Res. 2018; 3(1): 45-49. doi: 10.18689/mjdr-1000110
Copyright: © 2018 The Author(s). This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Skin is not a preferred target organ for cancer metastasis, nevertheless involvement of skin by metastatic deposits is a poor prognostic sign. It usually results from lymphatic embolization, hematogenous or contiguous spread and also by iatrogenic implantation of malignant cells following surgical procedures. Breast carcinoma is the second most common cancer in women and the most common tumor leading to the appearance of skin metastasis. Cutaneous metastasis from breast carcinoma has variety of clinicopathological features which usually create a dilemma for primary care clinicians as well as dermatologists. Therefore, a high index of suspicion along with detailed history, proper clinical examination and cytological or histopathological confirmation is required to make its definite diagnosis and to improve the outcome of such patients. We describe a case of a 50-year-old Indian woman, who presented with a solitary nodular lesion on the anterior chest wall which was initially clinically diagnosed as an infected sebaceous cyst but later on turned out to be a case of cutaneous metastasis from an asymptomatic and neglected left breast carcinoma.
Keywords: Breast Cancer; Cutaneous Metastasis; Sebaceous Cyst; Carcinoma Erysipeloides.
Introduction
Cutaneous metastasis is an unusual clinical finding, which is rarely encountered in clinical practice. It occurs in 0.7 to 10.4% of all cancer patients and represents only 2% of all skin neoplasms [1]. The clinical presentations and findings in a case of cutaneous metastasis are quite variable and subtle, as a cutaneous metastatic lesion not only mimics a benign skin lesion or a primary skin tumour, but it can also occur as an initial manifestation of an undetected internal malignancy and can be the first sign of dissemination or recurrence of an already diagnosed tumour [2, 3]. Therefore, early recognition of this rare entity is necessary as it alters the staging of a malignant disease, with the consequent therapeutic and prognostic implications; nevertheless, it is generally indicative of a poor prognosis. Among various cancers occurring in women, cutaneous metastasis is most frequently seen with breast cancer rather than with any other visceral malignancies. We herein report one such case of a solitary nodular anterior chest wall cutaneous metastasis from an occult as well as neglected case of left breast carcinoma in a 50-year-old Indian woman which masqueraded as an infected sebaceous cyst and created a diagnostic conundrum with therapeutic delay.
Case Report
A 50-year-old illiterate Indian woman presented to the surgical outpatient department with a 6-month history of a nodular lesion on the anterior chest wall. She also complained that this lesion was progressively increasing in size since last 15 days and it was associated with mild pain. There was no history of fever but she had weight and appetite loss. Her past, personal, medical, family history was insignificant. Her physical examination revealed a solitary, round-oval, erythematous left side anterior chest wall nodule which measured 2.5 x 2 cm in size. It was firm in consistency, slightly mobile and tender on palpation. There was local rise in the temperature over the lesion and the surrounding skin was normal (Figure 1).
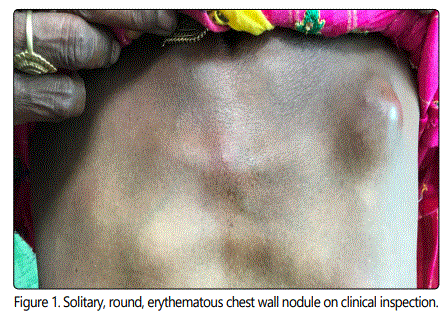
A clinical diagnosis of an infected sebaceous cyst was made and the patient was advised cytological correlation of the clinical diagnosis. Fine Needle Aspiration Cytology (FNAC) was performed. May Grunwald Giemsa (MGG) stained smears were cellular and showed malignant cells which were mainly dispersed singly and few were arranged in sheets, clusters and acinar pattern, against a lipoproteineceous background. Individual tumor cell was large with high nuclear to cytoplasmic ratio, moderate pale cytoplasm and had nucleus with coarse chromatin and prominent nucleoli. Numerous mitotic figures were also seen (Figure 2).
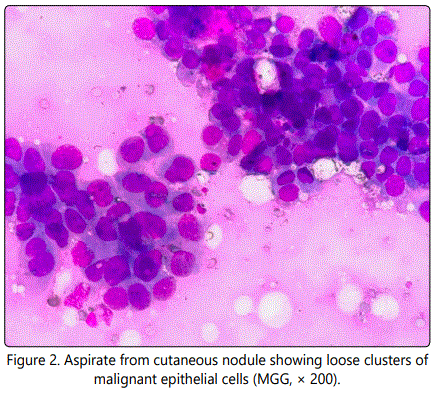
Based on these cytomorphological findings, a diagnosis of cutaneous deposits from an unknown primary tumor was made. Considering this cytological diagnosis, a detailed clinical examination as well as metastatic work-up was conducted, revealing a lump in the left breast. The lump was located in the upper inner quadrant and measured 3 x 3 cm in size. It was hard in consistency, irregular in shape, non-tender and freely mobile over the chest wall and the skin above. The overlying skin was normal. There was no nipple or areolar discharge or any cervical/axillary lymphadenopathy (Figure 3).
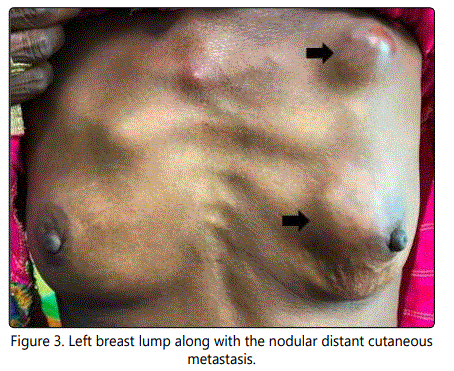
On further enquiring about the lump, the patient stated that she had this lump since last 20 years, initially it was small in size and recently it had increased in size, however, she never approached any doctor for it, as according to her she never had any problems pertaining to the breast lump. The rest of her general as well as all other systemic examinations were within normal limits. Routine hematological, biochemical, serological, and radiological investigations including abdominal ultrasonography (USG) were within normal limits. Mammography revealed an ill-defined mass with microcalcifications in the upper inner quadrant of the left breast. FNAC done from the left breast lump revealed features of infiltrating ductal carcinoma (IDC), which were similar as that of cutaneous deposits. Core needle biopsy from the breast lump showed features of IDC, with cells being estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor-2 (HER2) negative on immunohistochemistry (IHC). A skin biopsy was also performed and it confirmed the diagnosis of cutaneous deposits from breast carcinoma (carcinoma erysipeloides). The patient was planned for surgery followed by chemotherapy; however she never returned back for the treatment and was lost to follow-up.
Discussion
Metastasis is one of the life threatening characteristics of malignant tumours, which incontrovertibly signals the existence of a systemic disease, carry a limited survival period, and thus represent a terminal stage of any malignancy [4]. Cutaneous metastasis is generally regarded as a rare occurrence, the true incidence of which is still unknown. However, the incidence appears to be higher nowadays due to the higher rates of biopsy and diagnosis rather than a true escalation in the incidence [5]. A meta-analysis reviewed 1080 cases of cutaneous metastasis in 20,380 cancer patients, and reported an estimated rate of 5.3% [6]. Cutaneous metastatic skin lesions are extensions of tumours to the skin, preferentially occurring in the skin overlying or proximal to the primary tumour. Any malignant tumour can spread to the skin. However, in practice, a direct relationship has been found between the frequency of different malignant tumours and the appearance of such lesions. In men, the most common malignant tumours that have a propensity to metastasize to the skin are lung carcinoma (24%), colorectal carcinoma (19%), melanoma (13%), oral squamous cell carcinoma (12%), kidney (6%), stomach (6%), and oesophagus (3%) whereas, in women, the most common tumours are breast cancer (69%), colorectal carcinoma (9%), melanoma (5%), ovarian carcinoma (4%) and uterine cervix (2%) [7, 8]. Cutaneous metastasis is the presenting sign of the disease in 37% of men and 6% of women [8]. Carcinoma of the breast is the most common cancer especially among Indian women replacing cervical cancer [9]. An estimated lifetime breast cancer risk of 13% has been reported in any women [6, 10]. The documented incidence of cutaneous involvement in breast cancer is as high as 23.9% [11]. The tumour cells reach the skin through several routes: direct invasion from underlying structures, extension through lymphatics and blood vessels via embolization, and accidental implantation following a surgery, such as mastectomy or reconstruction. Cutaneous metastasis in breast cancer generally occurs late i.e. usually in the first three years after the initial diagnosis [12-14], however it can be diagnosed prior as well as concurrently with the primary tumour or it might be the presenting sign of an undiagnosed, asymptomatic breast cancer as seen in our case. The patient in the present case developed breast lump initially for which she did not seek any medical advice and later on, after almost two decades she presented with both the cutaneous metastasis and breast cancer.
Cutaneous metastasis due to breast cancer has several clinical and pathological features which warrants the attention from treating physicians. The highest incidence of cutaneous metastasis from breast cancer is seen between 50 to 70 years of age group. The most common presentation is in the form of rapidly growing, round to oval, single nodule or multiple nodules of variable sizes with firm to elastic consistency. Their color is similar to the normal adjacent skin but occasionally, they may ulcerate. The anterior chest wall, upper abdomen, scalp, contralateral breast and scar sites are the most frequent sites to be involved. The less common locations are the back, neck, upper arms, and lower abdomen; and rarely on the buttocks, perianal region, lower extremities, and eyelids [15]. Other clinical manifestations include carcinoma erysipelatoides, en cuirasse, telangiectatic, alopecia neoplastica, zosteriform and palpebral nodules [10, 15]. In a retrospective review by Mordenti et al. [16], 164 cases of skin metastasis from breast carcinoma were studied. Skin papules and/or nodules were found in 80% of patients, telangiectatic carcinomas in 11.2%, erysipeloid carcinomas in 3%, carcinoma ‘en cuirasse' in 3%, alopecia neoplastica in 2% and a zosteriform type in 0.8% of patients. Our patient presented with carcinoma erysipeloides, which is a relatively rare variant of metastatic disease and is characterized by skin metastasis with inflammatory skin changes and histological invasion of dermal lymphatics. It is usually associated with intraductal breast carcinoma [17]. This entity was first described by Lee and Tannenbaum in 1924 and was given its current name in 1931 by Rasch [18]. It is usually a diagnostic clue of breast cancer dissemination, and it rarely arises from any other internal malignancies. Although it mimics erysipelas, however, there is absence of fever and chills along with negative bacterial cultures and absence of leukocytosis. The pathogenesis behind this clinical picture is due to the spread of the metastatic cells along the subepidermal and subcutaneous lymphatics leading to blockade of the lymph ducts. The prognosis of such patients with this presentation is usually grave [19]. Nevertheless, over all, the clinical appearance of cutaneous metastasis can mimic other benign dermatologic lesions also, such as erythema annulare, contact dermatitis, tinea infections, erysipelas, cellulitis, and cutaneous mucinosis [20].
The diagnosis of cutaneous metastasis is mainly based on a thorough medical history and physical examination as sometimes the tumor may evade the radiological studies, cytology and histopathological examination of biopsy specimen. Nevertheless, a collaboration of all these modalities is usually required in reaching the definite diagnosis. Imaging studies especially USG, has often been implicated to rule out any underlying malignancy, as there are many dermal lesions which are usually benign and extra-parenchymal in location, and though not specific to the breast, can create a diagnostic dilemma. These lesions are epidermal inclusion cyst/ sebaceous cyst, vascular tumours, lipoma, infections, scars and fat necrosis [21, 22]. However, USG is not reliable in detecting cutaneous metastatic lesions from breast carcinoma as in most of the cases it fails in differential diagnosis [23]. Recently, the role of 2-deoxy-2-[fluorine-18] fluoro-D-glucose positron emission tomography (18F-FDG PET) in determining these lesions has been significant. Manohar et al. [24] have detected cutaneous metastasis from an asymptomatic carcinoma breast incidentally during 18F-FDG PET scanning while Borkar et al. [25] have also reported another similar case which was detected by this dynamic imaging technique. However, they have also documented that PET may understate tumours in some cases, and small metastatic lesions can be missed. Because of these limitations radiology cannot be relied upon for the definite diagnosis of cutaneous metastasis. Hence, cytological or histopathological confirmation is necessary in most of the cases to substantiate the clinical diagnosis.
Many researchers have documented that FNAC has been a boon for an early diagnosis of subcutaneous/cutaneous nodules as it is not only a rapid, cost-effective diagnostic tool but it is also an excellent non-invasive method because in the presence of characteristic cytomorphology, it obviates the need for more invasive procedures [26-28]. Ancillary tests like immunocytochemistry and formation of cell block/ button followed by histopathological examination and IHC have broadened the utilization of FNAC for definite diagnosis in many tumors [29, 30]. In our case too, FNAC played a pivotal role in clinching the diagnosis. On the other hand, histopathological examination of incisional or excisional skin biopsy is essential for a final diagnosis. It mostly reveals the same characteristics as the tumor of origin, however, in some cases metastatic lesion can be more anaplastic in appearance and exhibit less differentiation [31]. Nevertheless, there are certain characteristics which are suggestive of cutaneous metastasis like the presence of neoplastic cells within the lymph and blood vessels, localization of the lesion in the deep reticular dermis and hypodermis, and the presence of neoplastic cells running along the bundles of collagen. They typically develop as round nodules located in the dermis or hypodermis, and are not usually in contact with the epidermis (Grenz zone). Fibrosis and inflammation are also evident in some cases [14]. Few authors have documented that it might be difficult to differentiate metastatic skin lesions from specific dermal structures and primary skin tumors; as these types of lesion frequently share similar morphological patterns on histopathology. In such cases, immunohistochemical markers play a significant role in differentiating these entities [21, 32, 33]. Most breast carcinoma metastasis express cytokeratins (CK) 7 and 19, ER, PR, androgen receptors, mammaglobin, Gross cystic disease fluid protein 15 (GCDFP-15), epithelial membrane antigen (EMA), carcinoembryonic antigen (CEA), CAM 5.2, Cathepsin D and E-cadherin while negative immunoexpression is observed for CK 20, vimentin, thyroidlung transcription factor (TTF-1), WT-1, DPC4 [3, 34, 35]. Few researchers have emphasized that there is an association between ER and the time for metastasis. Primary tumors with positive immunoexpression for ER have significantly longer disease-free periods than ER negative neoplasms, thus contributing to the late appearance of the lesions [12, 14]. Cho et al. [36] have also mentioned in their case series that there is a difference in the metastatic pattern and onset of skin metastasis according to the breast cancer subtypes. In their study two cases of HER2 positive breast cancer showed only skin metastasis after immediate postoperative period and rapid clinical response to targeted therapy. Another two cases of triple negative breast cancer revealed thyroid and lung metastasis in addition to skin metastasis, and their response of cytotoxic chemotherapy was not definite. The other hormone positive breast cancer exhibited skin metastasis only, with a longer, slower, less progressive pattern than other subtypes. These studies highlight that the IHC evaluation of lesion biopsies not only has diagnostic and therapeutic implications but it also plays a pivotal role in understanding the genesis behind the association between the skin metastasis and primary breast cancer.
Most cases of cutaneous metastasis are usually detected at the end stage of any malignancy. Nevertheless, the recognition of cutaneous breast carcinoma metastasis often dramatically alters the therapeutic plans, especially when metastasis depicts persistence of cancer originally thought to be cured. The therapeutic approach is mainly based on appropriate management of the primary tumour, as most skin lesions with or without soft tissue infiltration are more likely the manifestations of systemic relapse and not readily manageable by local treatment, such as surgery and/or radiation therapy, even in patients with only skin lesions [37, 38]. However, palliative care comprising of adequate management of the pain, pruritis, bleeding, crusting, secondary super infection etc., is given if lesions are asymptomatic and the primary cancer is untreatable while chemotherapy can be used when skin metastatic lesions are large or show rapid progression, or when they are unresponsive to hormone therapy [39, 40].
The prognosis of a patient with cutaneous metastasis depends primarily on the type of underlying primary tumour and its response to treatment. In most of the cases, cutaneous metastasis from breast carcinoma usually occurs in advanced stages and is poorly amenable to treatment [3]. However, recent advances in chemotherapy have considerably increased survival period in all cancer patients and authors have reported a better survival in cases of breast cancer in comparison to other malignancies [41].
Conclusion
Cutaneous metastasis can present as a distressing sign of tumour recurrence or can be the first clinical manifestation of an occult tumour. Awareness about this entity and active screening of patients with a detailed medical history, complete physical examination and other relevant investigations like imaging studies, FNAC and histopathology along with IHC are paramount for clinching early diagnosis and instituting appropriate therapy, so as to increase the life expectancy of such patients.
Funding Source
No funding was secured for this case study.
Financial Disclosure
The authors have no financial disclosures relevant to this article.
Conflict of Interest
The authors have no conflicts of interest to disclose.
References