Review Article
Calculation of the ν4 (NH4+) IR Mode Frequency and the Damping Constant (FWHM) close to the Phase Transitions in NH4ZN(HCOO)3 and ND4ZN(DCOO)3
1Lapseki İÇDAŞ ÇİB MTAL High School, Çanakkale, Turkey
2Department of Physics, Middle East Technical University, Ankara, Turkey
3Department of Physics, Çanakkale 18 Mart University, Çanakkale, Turkey
*Corresponding author: Hamit Yurtseven, Professor, Department of Physics, Middle East Technical University, Ankara, Turkey, E-mail: hamit@metu.edu.tr
Received: February 28, 2019 Accepted: September 13, 2019 Published: September 23, 2019
Citation: Kurt A, Yurtseven H, Kurt M. Calculation of the ν4 (NH4+) IR Mode Frequency and the Damping Constant (Fwhm) close to the Phase Transitions in NH4ZN(HCOO)3 and ND4ZN(DCOO)3. Int J Chem Res. 2019; 1(2): 46-50. doi: 10.18689/ijcr-1000107
Copyright: © 2019 The Author(s). This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Temperature dependence of the IR frequency and the damping constant (FWHM) of the ν4(1440 cm-1) NH4+and ν4(1084 cm-1) ND4+ modes, are calculated for the metal formate frameworks (MOFs) of NH4Zn(HCOO)3 and ND4Zn(DCOO)3, respectively, by using the experimental data from the literature. By assuming the IR frequency of the ν4 mode as an order parameter, its temperature dependence is calculated close to the phase transition (TC=191 K) in the NH4Zn(HCOO)3 and ND4Zn(DCOO)3 by the molecular field theory. The temperature dependence of the damping constant of the ν4 (NH4+) IR mode is also calculated by means of the pseudospin-phonon coupled (PS) and the energy fluctuation (EF) models for these metal formate frameworks. The damping constant due to both models (PS and EF) is fitted to the observed FWHM data of the ν4 (NH4+) IR mode from the literature at various temperatures close to TC in the MOFs studied.
Our results show that the molecular field theory is adequate for the temperature dependence of the IR frequency and also the PS model explains the observed behaviour of the FWHM for the ν4(NH4+) mode in NH4Zn(HCOO)3 and ND4Zn(DCOO)3.
Keywords: IR frequency; Damping constant (FWHM); Molecular field theory; Pseudospin-phonon coupled (PS) model; MOFs.
Introduction
Metal formate frameworks (MOFs) with the ammonium cations, NH4M(HCOO)3 for M=Mn, Co and Ni compounds have been studied extensively by using X-ray diffraction, dielectric, differential scanning calorimetry (DSC) and magnetic measurements [1-5]. In particular, infrared (IR) and Raman bands in these structures have been assigned [3,4] in order to investigate both temperature- and pressure- induced phase transition mechanism including order-disorder, displacive transitions and H-bonds studies [6-9]. It has been pointed out that H-bonds and ordering of the ammonium ions play an important role in the mechanism of the phase transition (order-disorder) and multiferroelectric behavior of this family of compounds at low temperatures [5]. Dielectric properties of NH4Zn(HCOO)3 and ND4Zn(DCOO)3, DSC study of the deuterated sample, temperature-dependent of Raman scattering and infrared (IR) studies of both compounds, high-pressure Raman scattering of ND4Zn(DCOO)3 and DFT calculations of both structures have been reported to investigate their structural phase changes [5]. From the DSC measurements of ND4Zn(DCOO)3 an anomaly occurs at around 191 K upon heating and 190 K upon cooling, which is similar as observed for other NH4M(HCOO)3 formates with M=Mn, Co, Fe, Ni, Zn and Mg [1-4], indicating a second order, reversible phase transition, as pointed out previously [5]. On the other hand, for NH4Zn(HCOO)3 the phase transition was reported to be 192 K in the heating mode [1,2] which shows that the isotope effect for this compound is very small [5]. Anomaly was attributed to the onset of a ferroelectric order in the low temperature phase and the ferroelectricity of NH4Zn(HCOO)3 was confirmed by the presence of a dielectric hysteresis loop [1,2,10], as indicated previously [5].
The temperature dependence of the Raman and IR bands of NH4Zn(HCOO)3 and ND4Zn(DCOO)3 regarding internal vibrations of the ammonium and formate ions, and the lattice vibrations can be studied, as observed experimentally [5] in terms of their frequency shifts and FWHM. In particular, the IR frequency of the ν4 (NH4+) mode decreases which can be associated with the order parameter and its FWHM increases as the temperature increases toward the transition temperature Tc from low temperature (ordered phase) for NH4Zn(HCOO)3 and ND4Zn(DCOO)3 as observed experimentally [5], so that the temperature dependence of the IR frequency and FWHM of this mode can be calculated for those compounds. The anomalous behaviour of the observed IR frequency and FWHM of the ν4 (NH4+) mode can then be explained for the order-disorder transition in NH4Zn(HCOO)3 and ND4Zn(DCOO)3 compounds on the basis of the calculations by using the models used in this study. This is the motivation of our study given here to investigate the mechanism of the phase transitions in the MOFs considered. For the IR frequency of the ν4 (NH4+) mode, we use the molecular field theory [11] by calculating the temperature dependence of the order parameter. For the FWHM of this mode, we use mainly pseudospin-phonon coupling (PS) model [12] and also energyfluctuation (EF) model [13] for the damping constant of NH4Zn(HCOO)3 and ND4Zn(DCOO)3.
Materials and Methods
Temperature-dependent raman spectra were obtained for NH4Zn(HCOO)3 and ND4Zn(DCOO)3, which were measured using a bruker FT100/S spectrometer with YAG:Nd laser excitation (1064 nm) and a helium-flow oxford crysostat as pointed out in the previous study [5]. The vibrational spectra of NH4Zn(HCOO)3 and ND4Zn(DCOO)3 were regarded as composed of internal vibrations of the ammonium and formate ions, and the lattice vibrations, in particular, the bending modes ν2 and ν4 of the ammonium cation were observed [5] near 1600-1720 and 1390-1460 cm-1 (1100-1300 and 1050-1150 cm-1) for NH4+(ND4+), respectively [14-16].
The temperature dependent changes in vibrational spectra, in particular, plots of frequencies and full width at the half-maximum (fwhm) values for a number of structural units were presented [5]. In the present study, temperature dependence of the bending modes of ν4 (1440 cm-1) NH4 and ν4 (1084 cm-1)ND4+ are given (Figure 1). Temperature-dependent Raman spectra [5] are analyzed to predict the frequencies and FWHM for the bending modes ν4 NH4+ and ν4 ND4+ of NH4Zn(HCOO)3and ND4Zn(DCOO)3, respectively in this study.

Calculations and Results
The order parameter S can be calculated below the transition temperature Tc by the molecular field theory [11] according to the relation

Regarding order–disorder transition in molecular crystals, the critical behaviour of the frequency can be associated with the order parameter close to the transition. In particular, ν4 (NH4+) infrared mode frequency is the driving mechanism of the order-disorder transition in the metal formate frameworks (MOFs) of the compounds NH4Zn(HCOO)3 and ND4Zn(DCOO)3. The temperature dependence of this IR mode can be considered as an order parameter in those MOFs. On that basis, we have related the IR frequency of the ν4 (NH4+) mode to the order parameter by considering the temperature dependence of the frequency as

according to the relation
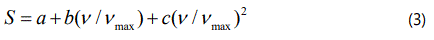
where a0, a1, a2, a, b and c are all constants. νmax denotes the maximum value of the IR ν4 (NH4+) frequency to normalize it (ν/νmax) since the order parameter (S) varies from 0 to 1. We assume here quadratic dependence of the order parameter on the frequency (Eq.3).
We first analyzed the temperature dependence of the ν4(NH4+) IR mode frequencies by using the experimental data [5] according to Eq.(2) with the parameters a0, a1 and a2 determined (Table 1). Calculated order parameter S (Eq.1) was then fitted to the observed frequency data (ν/νmax) by using Eq. (3) with the parameters a, b and c below Tc (Table 1). This analysis and calculation of the IR ν4(NH4+) mode were performed for the NH4Zn(HCOO)3 and its deutero compound of ND4Zn(DCOO)3 with the IR frequencies of 1440 and 1084 cm-1, respectively. Above Tc, since S=0 (Eq.1) the observed IR frequency data [5] were analyzed by using Eq.(2) with the parameters a0, a1 and a2 within the temperature intervals considered, as given in table 2. Figures 2 and 3 give the calculated wave numbers of 1440 and 1084 cm-1 for the ν4(NH4+) IR mode, with the observed data [5] as a function of temperature for the compounds of NH4Zn(HCOO)3 and ND4Zn(DCOO)3 respectively.
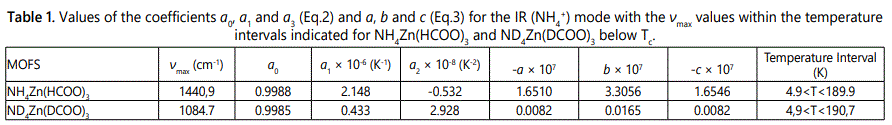
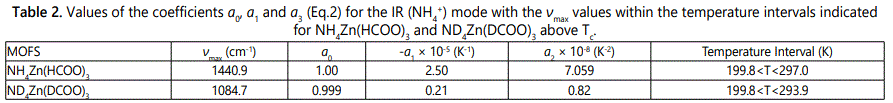


Temperature dependence of the damping constant (FWHM) can also be evaluated by using the ν4 (NH4+) IR mode frequency as an order parameter for NH4Zn(HCOO)3 and ND4Zn(DCOO)3. For this calculation, the pseudospin-phonon coupled (PS) model [12] was employed according to the relation
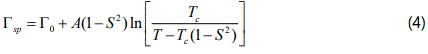
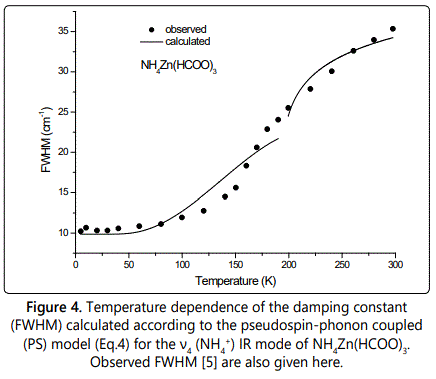
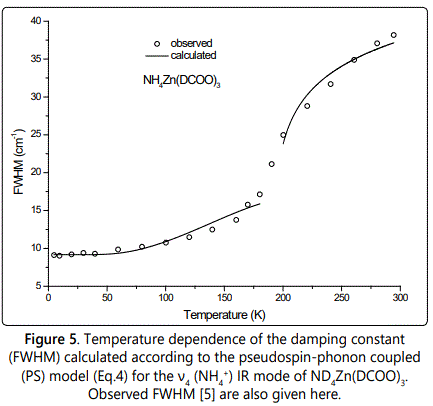
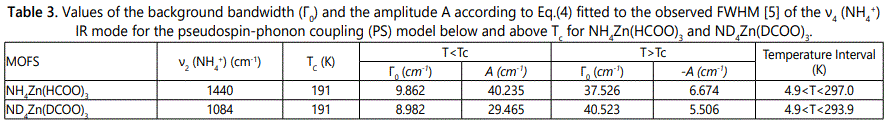
where Γ0 represents the background damping constant ΓSP due to the coupling between pseudospin (PS) of the (NH4+) ions and phonon in those MOFs and A is the amplitude. In Eq.(4) we used the IR ν4 (NH4+) frequency as an order parameter S. This calculation of ΓSP was carried out below (S≠0) and above (S=0) the transition temperature TC. Figures 4 and 5 give our calculated FWHM (damping constant ΓSP) according to Eq.(4) by using the 1440 and 1084 cm-1 IR frequencies calculated (Eqs.1 and 3) of the ν4 (NH4+) IR mode for NH4Zn(HCOO)3 and ND4Zn(DCOO)3 compounds, respectively, with the observed FWHM of those modes [5]. Table 3 gives the values of Γ0 and A for both IR modes (1440 and 1084 cm-1).
Discussion
The IR frequency of the ν4 (NH4+) mode for the MOFs of NH4Zn(HCOO)3 (1440 cm-1) and ND4Zn(DCOO)3 (1084 cm-1) was calculated as an order parameter S from the molecular field theory (Eq.1) through Eqs.(2) and (3) at various temperatures (Figures 2 and 3) as stated above. Our fits are reasonably good indicating that the ν4 (NH4+) IR mode can be considered as an order parameter and it can be associated with the mechanism of the order-disorder transition in those MOFs. In a wide temperature range below the transition temperature (TC≈191 K), we find that the molecular field theory (Eq.1) describes adequately the observed behaviour of the ν4 (NH4+) IR mode frequency. Close to TC, the temperature dependence of the order parameter S is given by the relation from the molecular field theory [11],

with the critical exponent β=1/2 for the order parameter according to the power-law S ∝∈β where the reduced temperature is ∈=(Tc-T)/Tc. Thus, in the vicinity of TC, the ν4(NH4+) IR frequency data can be analyzed by the power-law formula and the β-value (different from 1/2) can be obtained for NH4Zn(HCOO)3 and ND4Zn(DCOO)3. This requires the IR frequencies measured accurately in a very narrow temperature interval around TC.
For the mechanism of the phase transition in NH4Zn(HCOO)3 and ND4Zn(DCOO)3, the effect of isotopic substitution can be investigated since it influences phase transition temperature and facilitates assignment of modes to the respective motions of atoms in the unit cell [5] in these compounds with the extensive H-bonds. It has been pointed out that temperature-dependent studies reveal a very weak isotopic effect on the phase transition temperature and that ordering of ammonium cations plays a major role in the mechanism of the phase transition [5]. In fact, for both compounds of NH4Zn(HCOO)3 and ND4Zn(DCOO)3, transition temperature is almost the same (≈191 K) and due to the reorientations of the NH4+(ND4+) ions the ordered phase occurs at low temperatures (below TC). This also explains the multiferroic behaviour of those compounds at low temperatures.
Regarding the temperature dependence of the damping constant ΓSP (FWHM) due to the pseudospin-phonon coupling for the ν4 (NH4+) IR mode, the pseudospin-phonon coupled (PS) model is satisfactory to describe the observed behaviour of the IR line widths of this mode below TC (Figures 4 and 5). Above TC, since the order parameter is zero (S=0) as predicted from the molecular field theory (Eq.1) without using any IR frequency, Eq.(4) was fitted to the observed FWHM data of this mode, as mentioned before. We also used the energy fluctuation (EF) model [13] according to the relation
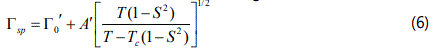
where Γ0′ and A′ are the background bandwidth and amplitude, respectively, as before. In contrast to the PS model, when Eq.(6) was fitted to the ν4 (NH4+) mode for the MOFs of NH4Zn(HCOO)3 and ND4Zn(DCOO)3 it was found that the agreement was not satisfactory. Most likely, this is due to the fact that the damping constant varies with the temperature as ΓSP∝(T-Tc)-1/2 with the critical exponent value of 1/2 (for example, when S=0 above TC). This then indicates that the EF model (Eq.6) can describe the observed behaviour (FWHM) in the vicinity of the TC (in a very narrow temperature range) for the ν4 (NH4+) IR mode of those MOFs. We note that for both models (PS and EF) due to the reorientation of the NH4+ ions which are ordered below TC, interaction between ammonium cations (spin) and the displacement of the metal formate framework (phonon) is considered for the mechanism of phase transitions in NH4Zn(HCOO)3 and ND4Zn(DCOO)3.
Increase in FWHM (Figures 4 and 5) indicates that the phase transition in NH4Zn(HCOO)3 and ND4Zn(DCOO)3 has an order-disorder character and it is associated with the rotational dynamics of the ammonium cations [3,4] as also pointed out previously [5]. Including phonon-phonon anharmonic interactions, from the temperature dependence of FWHM of the ν4 (NH4+) modes the activation energy was calculated in the thermal activated reorientational processes as 86 and 93 meV for NH4Zn(HCOO)3 and ND4Zn(DCOO)3, respectively [5]. This shows that if the potential curve for the motion of ND4+ equals that of NH4+, the activation energy for the reorientation increases by the deutration due to the increase of the moment of inertia [17], as also pointed out previously [5]. This is also an indication of the interaction between the ammonium cations and anionic framework, which is weakly affected by the deuteration [5].
Conclusions
The infrared (IR) frequencies and the line width (damping constant) of the ν4 (NH4+) mode were calculated as a function of temperature close to the order-disorder transition in the MOFs of NH4Zn(HCOO)3 and ND4Zn(DCOO)3. The IR frequency of this mode as an order parameter and its damping constant (FWHM) were calculated from the molecular field theory and the pseudospin-phonon coupling (PS) model, respectively.
Our results show that the observed behaviour of the IR frequency and the line width for the ν4 (NH4+) mode can be described satisfactory by the models used for the compounds studied here.
References
- Wang Z, Zhang B, Inoue K, et al. Occurrence of a Rare 49 ·66 Structural Topology, Chirality, and Weak Ferromagnetism in the [NH4][MII(HCOO)3] (M=Mn, Co, Ni) Frameworks. Inorg Chem. 2007; 46(2): 437-445. doi: 10.1021/ic0610031
- Xu GC, Zhang W, Ma XM, et al. Coexistence of Magnetic and Electric Orderings in the Metal–Formate Frameworks of [NH4][M(HCOO)3]. J Am Chem Soc. 2011; 133(38): 14948-14951. doi: 10.1021/ja206891q
- Mączka M, Pietraszko A, Macalik B, Hermanowicz K. Structure, Phonon Properties, and Order–Disorder Transition in the Metal Formate Framework of [NH4][Mg(HCOO)3]. Inorg Chem. 2014; 53(2): 787-794. doi: 10.1021/ic4020702
- Zhang R, Xu GC, Wang ZM, Gao S. Phase transitions, prominent dielectric anomalies, and negative thermal expansion in three high thermally stable ammonium magnesium-formate frameworks. Chemistry. 2014; 20(4): 1146-1158. doi: 10.1002/chem.201303425
- Mączka M, Kadłubański P, Freire PTC, et al. Temperature- and PressureInduced Phase Transitions in the Metal Formate Framework of [ND4][Zn(DCOO)3] and [NH4][Zn(HCOO)3]. Inorg Chem. 2014; 53(18): 9615-9624. doi: 10.1021/ic501074x
- Nibbering ETJ, Dreyer J, Kühn O, Bredenbeck J, Hamm P, Elsaesser T. Vibrational dynamics of hydrogen bonds. In: Kühn O, Wöste L (eds). Analysis and Control of Ultrafast Photoinduced Reactions. Chemical Physics. 2007; 87: 619-687.
- Carabatos-Nédelec C, Becker P. Order–disorder and structural phase transitions in solid-state materials by Raman scattering analysis. J Raman Spectrosc. 1997; 28: 663-671. doi: 10.1002/(SICI)1097-4555(199709)28:9<663::AID-JRS157>3.0.CO;2-L
- Cummins HZ, Levanyuk AP. Light Scattering Near Phase Transitions. North-Holland Publishing Company. 1983: 682.
- Guo M, Cai HL, Xiong RG. Ferroelectric metal organic framework (MOF) Inorg Chem Commun. 2010; 13(12): 1590-1598. doi: 10.1016/j.inoche.2010.09.005
- Brout R. Phase Transitions. Chapter 2. Benjamin WA Publisher. New York, USA. 1965.
- Lahajnar G, Blinc R, Zumer S. Proton spin-lattice relaxation by critical polarization fluctuations in KH2PO4. Phys Cond Matte . 1974; 18(4): 301-316.
- Schaack G, Winterfeldt V. Temperature behaviour of optical phonons near Tc in triglycine sulphate and triglycine selenate. Ferroelectrics. 1977; 15(1): 35-41. doi: 10.1080/00150197708236718
- Hadrich A, Lautié A, Mhiri T. Vibrational study of structural phase transitions in (NH4)2HPO4 and (ND4)2DPO4. J Raman Spectrosc. 2000; 31(7): 587-593. doi: 10.1002/1097-4555(200007)31:7<587::AID-JRS581>3.0.CO;2-7
- Kruger A, Heyns AM. A Raman and infrared study of (NH4)2ZrF6. Vib Spectrosc. 1997; 14(2): 171-181. doi: 10.1016/S0924-2031(96)00072-0
- Fredrickson RL, Decius JC. The Raman spectrum of the ordered phase of NH4Cl and ND4Cl: Dipole and polarizability derivatives. J Chem Phys. 1977; 66: 2297. doi: 10.1063/1.434291
- Asaji T, Ishizaka T. Effect of Deuteration on Ammonium Motion and Structural Phase Transition Studied by Nuclear Quadrupole Resonance. Z Naturforsch. 2000; 55: 83-89.


