Mini Review Article
Left Atrial Appendage morphology and Flow assessment by Cardiovascular Magnetic Resonance Imaging
Brody School of Medicine, East Carolina University, Greenville, NC, USA
*Corresponding author: Constantin B Marcu, Brody School of Medicine, East Carolina University, Greenville, NC, USA, E-mail: marcuc@ecu.edu
Received: October 11, 2017 Accepted: October 28, 2017 Published: November 3, 2017
Citation: Mehra S, Marcu CB. Left Atrial Appendage morphology and Flow assessment by Cardiovascular Magnetic Resonance Imaging. Madridge J Cardiol. 2017: 2(1): 27-29. doi: 10.18689/mjc-1000108
Copyright: © 2017 The Author(s). This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Patients with atrial fibrillation (AF) and atrial flutter (AFL) are at increased risk for thromboembolic events due to left atrial thrombi. Transesophageal echocardiogram (TEE) is the procedure of choice to exclude left atrial thrombi before performing cardioversion or ablation procedures in these patients. TEE is an invasive procedure and not without risks. Cardiac magnetic resonance imaging (CMR) is a non-invasive alternative for patients in whom TEE cannot be safely performed to exclude left atrial thrombi.
Keywords: Atrial fibrillation; Atrial flutter; Thromboembolic; Transesophageal echocardiogram; Cardioversion; Cardiovascular magnetic resonance.
Abbreviations: AF: Atrial Fibrillation; AFL: Atrial Flutter; TEE: Transesophageal Echocardiogram; CMR: Cardiovascular Magnetic Resonance imaging; LAA: Left Atrial Appendage; CCT: Cardiac Computed Tomography; ECG: Electrocardiogram; IR: Inversion Recovery.
Introduction
Patients with chronic atrial flutter (AFL) are at increased risk of thromboembolic events, which may occur after electrical cardioversion or spontaneous conversion to sinus rhythm. Schmidt, et al evaluated 139 patients with AFL and found spontaneous echo contrast in 28% cases and thrombi in 1% cases. Decreased atrial mechanical activity and reduced blood flow velocities during episodes of AFL or AF may lead to left atrial thrombi formation, and consequent peripheral embolization. The main location for left atrial thrombus formation is the left atrial appendage (LAA). Patients with increased thrombogenic milieu such as with decreased left ventricular ejection fraction (<40%), diabetes mellitus or arterial hypertension should generally undergo a TEE to exclude LAA thrombi before attempts at cardioversion [1-3]. TEE is the procedure of choice for diagnosis of LAA thrombus, with high diagnostic accuracy- sensitivity of 100% and specificity of 99% [4]. However, TEE is a semi-invasive procedure and with some associated risks [5]. The overall complication rates for TEE vary from 0.18 to 2.8%. Common complications include lip injury (13%), hoarseness (12%) and dysphagia (1.8%). More serious complications including esophageal perforation (<0.01%), major bleeding (<0.01%) and death (<0.01-0.02%) are much rarer. The risk of complications is higher in patients with esophageal pathology such as strictures, esophageal diverticulum or esophageal tumor. TEE is contraindicated in such situations [6]. Cardiovascular magnetic resonance Imaging (CMR) has been proposed as an alternative to TEE for evaluation of left atrial and LAA thrombi before contemplating cardioversion or ablation in patients with AF or AFL.
Clinical Presentation
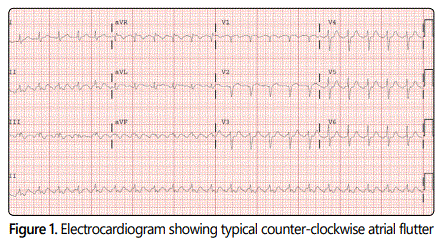
We present the case of a 72-year-old male with previous history of paroxysmal AF on anticoagulation with apixaban who presented to our emergency room with palpitations and was found to be in AFL [Figure 1]. Patient received intravenous diltiazem and digoxin, orally, for rate control. Plan was to perform TEE to rule out presence of LAA thrombus followed by AFL radiofrequency ablation. The patient had a history of a Zenkerʼs diverticulum of the esophagus which was confirmed by endoscopy. TEE was contraindicated due to presence of diverticulum. A CMR with and without gadolinium contrast using a 1.5 T magnet was performed in an attempt to visualize the left atrium and LAA and to rule out LAA thrombus.
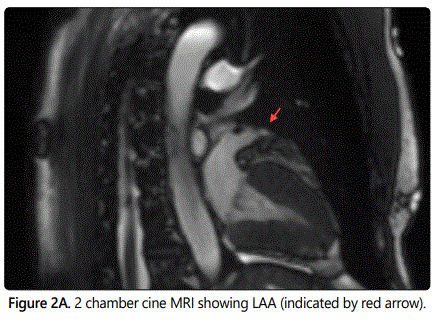
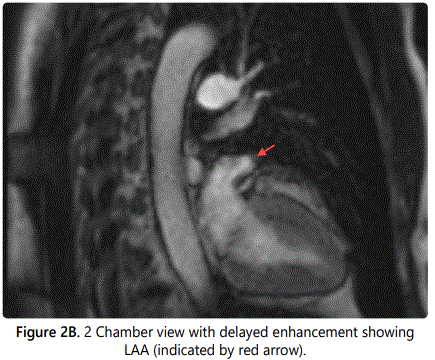
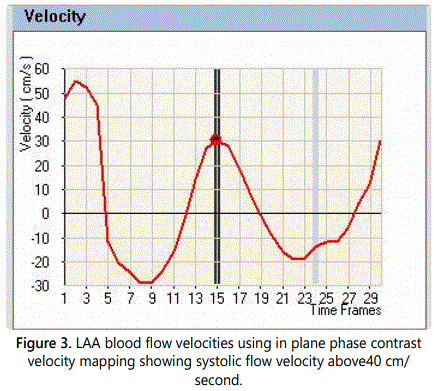
CMR showed no evidence of filling defects in the LAA to suggest presence of thrombus on both cine and delayed gadolinium contrast enhanced sequences [Figures 2A and B]. LAA blood flow velocities were above 40 cm/s on through plane phase contrast velocity mapping sequences. [Figure3] The left atrium was normal in size. Subsequently patient underwent successful AFL ablation. Patient continued systemic anticoagulation with apixaban for primary prevention of thromboembolic events.
Discussion
TEE is the procedure of choice for ruling out presence of LAA thrombus in selected patients with AF or AFL before cardioversion or ablation procedures. When TEE is contraindicated, such as in the case of our patient, CMR or cardiac computed tomography (CCT) could be considered as alternative imaging methods. CCT has high spatial resolution but requires administration of iodinated contrast and ionizing radiation. CMR has excellent spatial and temporal resolution without use of ionizing radiation. Barkhausen, et al compared the various CMR techniques with diagnostic accuracy of transthoracic and trans-esophageal echocardiography and found CMR imaging more sensitive for detection of left ventricular thrombus. Variation of inversion times could be used to improve contrast between thrombi and myocardium. The use of gadolinium contrast helped to characterize thrombi. ECG- triggered 3D inversion-recovery turbo FLASH sequence used in this study allowed for full cardiac coverage in a single breath hold but was limited by artifacts such as in cases of atrial fibrillation or other cardiac arrhythmias [7].
Delewi, et al compared the diagnostic accuracy of TTE with CMR in patients with ST-elevation myocardial infarction undergoing primary percutaneous intervention and found that routine TTE had only 21-24% sensitivity and 95-98% specificity compared to CMR for the detection of LV thrombi. TTE also had a higher intra- and inter observer variation of detection of LV thrombus compared to CMR [8].
Ohyama, et al evaluated 50 patients with non-rheumatic AF and a prior history of cardio embolic stroke who underwent same day TEE using a 5 MHz multi-plane probe and CMR using a 1.5 T whole body magnet, for thrombus detection in the LAA. Both double and triple–inversion recovery (IR) sequences were used during CMR. The LAA was visualized clearly in all 50 patients. CMR detected well-circumscribed LAA filling defects, which were isointense to the left ventricular myocardium on double-IR sequence and relative high intensity in the triple-IR sequence. TEE confirmed presence of LAA thrombi in 16 of the 19 patients with suspected LAA filling defects seen on CMR. The study inferred high concordance between CMR and TEE for detection of LAA thrombus [9].
Conclusion
CMR is a non-invasive modality which can detect the presence of LAA thrombus in selected patients with AF or AFL. CMR can serve as an alternative to TEE in patients with either contra-indications to TEE, for patients refusing TEE or when TEE attempts are not successful.
CMR image quality can be degraded by rapid and irregular patient heart rates or poor breath-hold techniques. Limited availability of cardiac imaging capable magnets and CMR specialists remain a limiting factor.
Conflicts of Interest: The authors declare None of the authors have any competing interests in the manuscript.
References
- Lanzarotti CJ, Olshansky B. Thromboembolism in chronic atrial flutter: is the risk underestimated? J Am Coll Cardiol. 1997; 30: 1506-11. doi: 10.1016/S0735-1097(97)00326-4
- Schmidt H, Von der Recke G, Illien S, et al. Prevalence of Left Atrial Chamber and Appendage Thrombi in Patients With Atrial Flutter and Its Clinical Significance. J Am Coll Cardiol. 2001; 38: 778-84. doi: 10.1016/S0735-1097(01)01463-2
- Manning WJ, Weintraub RM, Waksmonski CA, et al. Accuracy of transesophageal echocardiographyfor identifying left atrial thrombi: a prospective, intraoperative study. Ann Intern Med. 1995; 123: 817-822. doi: 10.7326/0003-4819-123-11-199512010-00001
- Hilberath JN, Oakes DA, Shernan SK, et al. Safety of Transesophageal Echocardiography. JASE. 2010; 23(11): 1115-1127. doi: 10.1016/j.echo.2010.08.013
- Barkhausen J, Hunold P, Eggebrecht H, et al. Detection and characterization of intracardiac thrombi on MR imaging. AJR Am J Roentgenol. 2002; 179(6): 1539-44. doi: 10.2214/ajr.179.6.1791539
- Delewi R, Nijveldt R, Hirsch A, et al. Left ventricular thrombus formation after acute myocardial infarction as assessed by cardiovascular magnetic resonance imaging. European Journal of Radiology. 2012; 81: 3900-3904. doi: 10.1016/j.ejrad.2012.06.029
- Ohyama H, Hosomi N, Takahashi T, et al. Comparison of Magnetic Resonance Imaging and Transesophageal Echocardiography in Detection of Thrombus in the Left Atrial Appendage. Stroke. 2003; 34: 2436-2439. doi: 10.1161/01.STR.0000090350.73614.0F


