Research Article
Impact of different organ failures on outcome of Acute Necrotizing Pancreatitis
1O.O. Shalimov National Institute of Surgery and Transplantology, Kyiv, Ukraine
2Bukovinian State Medical University, Chernivtsi, Ukraine
35th City Clinical Hospital, Minsk, Belarus
*Corresponding author: Oleksandr V Rotar, Associate Professor, Department of General Surgery, Bukovinian State Medical University, Chernivtsi, Ukraine, Tel: +380-(50) 1943590, E-mail: oleksandr.rotar@gmail.com
Received: January 29, 2018 Accepted: March 26, 2018 Published: March 30, 2018
Citation: Khomiak IV, Rotar OV, Nazarchuck MF, et al. Impact of different Organ Failures on Outcome of Acute Necrotizing Pancreatitis. Madridge J Surg. 2018; 1(2): 29-33. doi: 10.18689/mjs-1000107
Copyright: © 2018 The Author(s). This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background: Acute necrotizing pancreatitis is potentially lethal inflammatory process. Recent researches established that presences of multiorgan failure together with pancreatic infection are major determinants of its mortality. But significance of different organs failure, especially intestinal dysfunction in course of acute necrotizing pancreatitis is still remaining unclear.
Objective: To determine frequency of multiorgan failure occurrence during acute necrotizing pancreatitis and estimate significance of intestinal failure on mortality of disease.
Methods: We performed a prospective observational cohort study of 113 patients with necrotizing pancreatitis which were admitted to single intensive care department. Patients were included in the current study if they fulfilled the inclusion criteria of signs of pancreatic necrosis and/or peripancreatic necrosis. The following parameters were collected for each episode of acute pancreatitis: length of hospital stay, in-hospital mortality, presence of organ failure and local complications.
Results: Persistent organ failure was diagnosed in 50 (44%) and transient - in 63 (56%) observations. Death appeared at 31 (27.4%) of patients: 28 (56%) with persistent and 3 (4.8%) with transient organ failure and system complications. Respiratory failure (67%) has been dominated in mortality structure, cardio-vascular (59%), renal (56%) and intestinal (54%) failures were met with equal frequency. Serum citrulline concentration reflected the level of intestinal injury.
Conclusions: Amount of organs in multi organ failure syndrome, respiratory, cardiovascular, renal and intestinal failures are independent factors of mortality in patients with acute necrotizing pancreatitis. Plasma citrulline concentration is simple and reliable marker for diagnosing of intestinal failure.
Keywords: Acute necrotizing pancreatitis; Organ failure; Intestinal failure; Hospital mortality.
Introduction
An increasing incidence of acute pancreatitis has been reported recently, with an estimated 21,000 new cases per year in the United States [1]. On the one hand, most of these cases are the mild form of pancreatitis, without severe gland injury and characterized with complete recovery. On the other hand, acute necrotizing pancreatitisis associated with increased morbidity and mortality. Assessment of the severity of the attack and predicting the course of the disease are important when determining the allocation of care for these patients or the need for transfer to a higher level care early in the disease process. Recent researches established that presences of multiorgan failure together with pancreatic infection are major determinants of mortality during acute necrotizing pancreatitis [2, 3]. The mortality rates in the absence of organ failure, in the presence of one organ failure, and in patients with multiorgan failure are 0%, 3%, and 47%, respectively [4, 5, 6]. The gastrointestinal system is considered critical to the development of organ failures, with bacterial translocation in intensive care unit patients supporting the concept of the gut having a role in multiorgan failure [7, 8]. According to the gut hypothesis of organ failure, the multisystemic disorders defining this syndrome could be the late consequences of a previous small bowel ischemia. Currently there is no validated marker of acute intestinal dysfunction in critically ill patients [9], where as initial determination of severity in acute necrotizing pancreatitis to evaluate organ failure and the complications of pancreatic necrosis as soon as possible is critical for the appropriate management and risk assessment in a clinical setting.
Materials and Methods
This prospective observational cohort study was performed at Regional Emergency Hospital, Chernivtsi, Ukraine, between March 2010 and September 2015. The study was conducted in accordance with the principles of the Declaration of Helsinki. The ethics review board of each participating hospital approved the study. Patients or their legal representatives gave written informed consent. During this period 708 patients with acute pancreatitis were admitted to clinic. Acute pancreatitis was diagnosed if a patient had more than two of the three following findings: typical abdominal pain (acute onset of a persistent and severe epigastric pain often radiating to the back), elevation of serum amylase and/or lipase levels above three times upper normal limit, and findings of imaging studies. Most of them had moderate interstitial form of disease and were successfully treated at general surgical departments. Patients were included in the current prospective study if they fulfilled the inclusion criteria of signs of pancreatic necrosis on contrast-enhanced computed tomography. Necrosis was diagnosed by lack of pancreas enhancement on contrast computed tomography, which was a sign of impaired or absent tissue perfusion. There were 132 cases of acute necrotizing pancreatitis. Patients were not enrolled to the study if any of the following criteria were present: a) age < 18 and > 80 years; b) recent surgical interventions; c) psychoses; d) pregnancy; e) previously history of chronic pancreatitis. After exclusion of abovementioned cases we enrolled to current study 113 patients with acute necrotizing pancreatitis. All of them were admitted to single intensive care department.
Severity of acute necrotizing pancreatitis was determined according to the recently revised Atlanta Classification [10]. Moderately severe acute necrotizing pancreatitis was defined by the presence of transient organ failure, local complications such as acute peripancreatic fluid collections and acute necrotic collections, or exacerbation of comorbid diseases. Severe acute necrotizing pancreatitis was diagnosed in case lasting of persistent organ failure for more than 48 h. Organ failure was defined for respiratory, cardiovascular, and renal system by modified Marshall scoring system [10]. Besides above organs dysfunction of neurological system, liver and intestine were estimated. Neurological failure was established by Glasgowcoma score. Hepaticdys function was evaluated according to Child-Pugh classification [11], grade B or C were criteria for diagnosis of liver failure. Intestinal injury was estimated by modified gastrointestinal failure score [12]. Three level of severity were defined: 1st -intolerance of enteralfeeding more than 3 days, depressed intestinal peristalsis, preserved intestinalgas evacuationability; 2nd-intestinal dysfunction: clinical and roentgenological signs of distended intestine with intraabdominal pressure 12-15 mm Hg, suppressed gas evacuation; 3rd-intestinal failure: clinic of paralyticintestinal obstruction with intraabdominal hypertension over 15 mm Hg, acute gastric and duodenal erosions with hemorrhage. Only 3rd grade of gastrointestinal injury was recognized as intestinal failure. Intra-abdominal pressure was measured via the bladder, with patients in the supine position, using the closed loop system repeated measurements technique [13]. Additionally, venous blood citrulline concentration was determined as indicator of intestinal epitheliocytes functional activity [14, 15].
After admission, patients received fluid resuscitation, and full laboratory investigations were performed on the first 3 days and in the 24 hours before any intervention. Nasojejunal enteral feeding was initiated if an oral diet was not tolerated after 48-72 hours after admission. If there were problems with nasojejunal intubation nasogastric tube feeding was started. Parenteral nutrition was only initiated when oral route was not tolerated or sufficient, but at least small amount of enteral feeding was present in all patients. In line with international guidelines, intervention was generally only performed in case of suspected or confirmed infection of pancreatic necrosis or peripancreatic necrosis alone. There was no routine fine-needle aspiration of peripancreatic collections. The decision to intervene was mostly based on clinical grounds (i.e., deterioration). Whenever possible, intervention was postponed until approximately four weeks after the onset of disease. In case of proven or suspected infection of pancreatic necrosis within four weeks after onset of disease, antibiotics were administered and intervention was postponed as long as possible. Some patients underwent emergency laparotomy early in the course of disease. The indication was severe clinical deterioration suspected to be caused by abdominal compartment syndrome, bowel ischemia, or perforation of a visceral organ. All patients were randomized on moderately severe and severe acute necrotizing pancreatitis groups. The following parameters were collected for each episode of acute necrotizing pancreatitis: length of hospital stay, in-hospital mortality, presence of organ failure, and local complications such as acute peritoneal fluid collection, pseudocyst, acute necrotic collection and walled-off pancreatic necrosis. Acute Physiology and Chronic Health Evaluation (APACHE)-II, Marshall scores, presence of organ failures and systemic inflammatory response syndrome (SIRS) were calculated daily during period of ICU treatment.
Statistics data were collected in a Microsoft Excel database. After completion of data collection, the database was imported into SPSS for Windows (SPSS 16.0., Chicago, IL, USA). Continuous variables were expressed as mean with standard deviation and compared using Student's t- test or Mann - Whitney test. Categorical variables were expressed as absolute numbers and proportions. Pearson's χ2test or Fisher's exact test was used for comparison of categorical variables. Logistic regression models were created to analyze the independent effects of organ failures on mortality. To define predisposing factors, the odds ratio and its 95% confidence interval were used as a binary parameter. Receiver operator curve (ROC) was generated to determine cutoff value of citrulline for estimation of intestinal failure. A P-value of < 0.05 was considered statistically significant.
Results and Discussion
All patients with acute necrotizing pancreatitis were belonged to two groups (table 1). According to recent revision of Atlanta classification [10] severe form (1st group) was established in 50 (44%) patients with persistent organ failure and local complication, moderate severe form (2nd group) was diagnosed in 63 (56%) persons with transient organ failure or local complications. APACHE-II and Marshall scores, hematocrit and creatinine level were significantly higher in patients of 1st group (p<0.05). There were no differences between groups according to age and frequency of local complications.
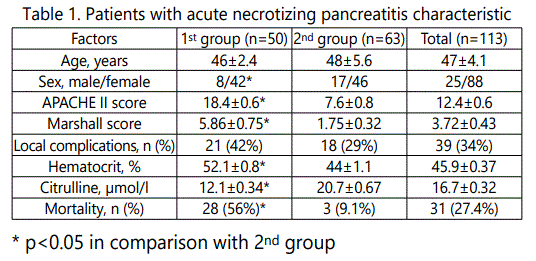
Death appeared at 31 (27.4%) cases from 113 patients. High hospital mortality was observed only among persons with severe form of acute necrotizing pancreatitis. Persistent organ failure occurred in 50 patients from 1st group, 28 (56%) of them died (table 2). Transient organ dysfunction appeared in 33 patients from 2nd group, there were 3 (4.76%) lethal cases. Intensive therapy, which included rigorous fluids resuscitation, hemodynamic and respiratory support, was ineffective in 17 patients, who have died at early phase of acute necrotizing pancreatitis (during first or second week of disease) due to pancreatic shock with multiple organ failure. During late phase of acute necrotizing pancreatitis (after two weeks from onset) there were 14 lethal cases as result of infection of pancreatic necrosis with development of sepsis and multiorgan failure. Amount of organs with dysfunction were significantly higher in deceased patients compared with survivors (3.48±0.81 and 1.33±0.44, respectively, p<0.05) as well as APACHE II score, age, hematocrit and creatinine level (table 2). Multivariate logistic regression analysis revealed that amount of organs in multiorgan failure were the main prognostic factor of death in patients with acute necrotizing pancreatitis (p<0.01), wherein each organ failure development significantly worsened outcome too. Respiratory failure had been dominated in mortality structure. Isolated respiratory failure was identified in 8 cases, 2 (25%) of them died. In patients with respiratory dysfunction accompanied by other organ failure mortality rate increased till 66.7-85.6%. Cardio-vascular (59%), renal (56%) and intestinal (54%) failures were met with equal frequency. Neurological and liver failures did not influence on patients survival. Intestinal failure(3rd level of intestinal injury score) was diagnosed in 21 (75%) among 28 deceased patients of 1st group, lasted more than 5 days and presented by signs of paralytic ileus, intra-abdominal compartment syndrome, acute gastric erosions and ulcers with bleeding. There venous citrulline concentration felt to 10.31±0.42 µmol/l during 48 hours after admission and was significantly lower (p<0.001) than in survived persons. In control group citrulline venous concentration was 36.8±0.43 µmol/l, in patients with moderate acute necrotizing pancreatitis it decreased till 20.7±0.67 µmol/l and in persons with severe acute necrotizing pancreatitis -felt almost three time (p<0.001, table2). Citrulline level below 12.5 µmol/l discriminated intestinal failure with sensitivity 0.904 and specificity 0.846 (AUC 0.905±0.0179, p=0.001, fig. 1). There was significant invert correlation (r= -0.643, p<0.05, fig. 2) between gastrointestinal injury score and citrulline concentration.
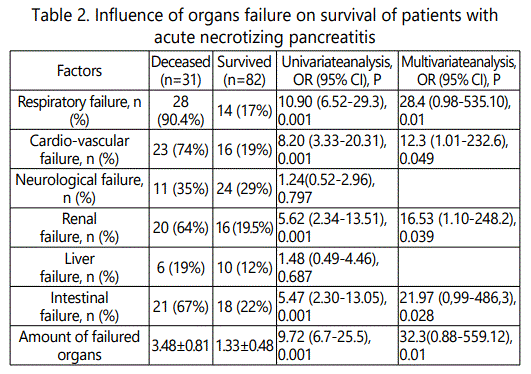
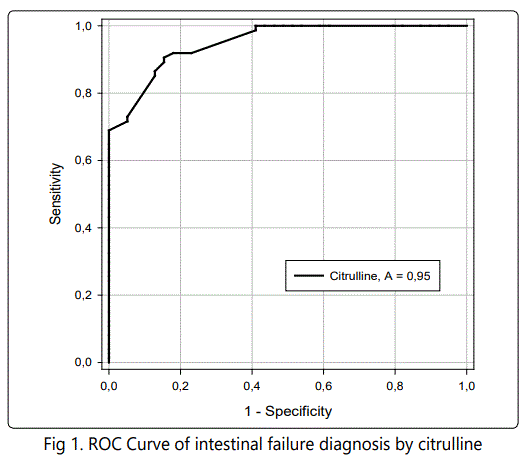
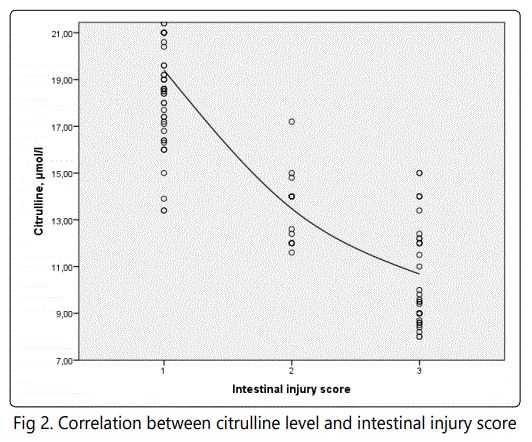
Our study supports the validity of main agenda of the 2012 revised Atlanta classification - importance of duration of organ failure for patient's prognosis. Mortality rate was more than ten times higher in patients with persistent organ failure (56%) comparing with transient (4.76%). Such relation was reliable as during early period of acute necrotizing pancreatitis either late phase. APACHE II score, age, hematocrit and creatinine levels were significantly higher in deceased patients but only organ failure amount has prognostic power on survival rate according to multivariate logistic regression. Besides validation of 2012 revised Atlanta classification we analyzed the influence of different organ failure on disease severity. Respiratory dysfunction occurred most frequently in patients with severe acute necrotizing pancreatitis and was followed by cardio-vascular (59%), renal (56%) and intestinal (54%) failures, which have similar impact on mortality rate. Recommended by 2012 Atlanta classification committee modified Marshall score as well as other widely accepted organ failure scores like SOFA, MODS, APACHE II are dealing with respiratory, cardio-vascular and renal failures but missing IF. Evaluating the intestine is difficult for two reasons: it is a deep organ, far from the mouth, anus, and abdominal wall, and critically ill patients are frequently not able to inform clinicians about a digestive complaint. This explains why its dysfunction may sometimes be occult or misdiagnosed, and the fact that it is not clearly integrated into the overall approach used to treat intensive care unit patients. However, even occult small bowel dysfunction may have clinical consequences on patient prognosis. The gut hypothesis of multiple organ failure was first put forward more than 20 years ago [7]. According to this hypothesis, the multisystemic disorders defining this syndrome could be the late consequences of a previous small bowel injury. If the intestinal failure could be evaluated and protected on admission, gutrelated sepsis will be reduced, and the mortality rate will fall down. Reintam et al. [12] found that 59% of critically ill patients have at least one gastrointestinal symptom during their stay in the intensive care unit and introduced gastrointestinal dysfunction score which was based on presence of feeding intolerance and intra abdominal hypertension. However, despite a welcome contribution to assessing intestinal failure, it was noted that feeding intolerance was a subjective measurement, and that intra abdominal hypertension was not specific to gastrointestinal dysfunction [13]. Although the gastrointestinal failure score proposed by Reintam et al. appeared to be a new prognostic tool to evaluate critically ill patients, it was not based on a pathophysiological model of gastrointestinal dysfunction. We chose to examine changes of citrulline concentration as a marker of enterocyte mass in patients with acute necrotizing pancreatitis. This amino acid is synthesized exclusively by small intestinal mucosal epitheliocytes thus it may serve as a measure of intestinal mucosal functional activity [15]. Recently it was established that plasma citrulline concentrations were able to reflect enterocyte mass in various small intestinal diseases including coeliac disease [16], short bowel syndrome [17], radiation enteritis [18] and Crohn disease [19]. In our research citrulline level decreased 1.5 times in patients with moderately severe pancreatitis and 3 times (p<0.05)-in patients with severe form, which was highly inverse correlated (r=-0.643, p<0.05) with clinical gastrointestinal failure score. In deceased persons its concentration dropped less than 10.31±0.42 µmol/l indicating critical level of intestinal failure due to deep injury of mucosal villi, desquamation of intestinal epithelia and high rate of bacterial and endotoxin translocation.
Conclusions
Amount of organs in multiorgan failure syndrome, respiratory, cardiovascular, renal and intestinal failures are independent factors of mortality in patients with acute necrotizing pancreatitis. Serum citrulline concentration below 12.5 µmol/l is simple and reliable marker for diagnosing of intestinal failure.
Acknowledgments
The authors are grateful to all contributors to this research especially the director of the private unitary enterprise "ARGOMEDA", Minsk, Belarus, Dr. Yaraslau Latushka and stuff of surgical and intensive care departments of Chernivtsi Emergency Regional Hospital, Ukraine for helping of performing research.
References
- Peery AF, Dellon ES, Lund J, Crockett SD, McGowan CE, et al. Burden of gastrointestinal disease in the United States: 2012 update. Gastroenterology. 2012; 143(5): 1179-1187. doi: 10.1053/j.gastro.2012.08.002
- Alsfasser G, Schwandner F, Pertschy A, Hauenstein K, Foitzik T, et al. Treatment of necrotizing pancreatitis: redefining the role of surgery. World J Surg. 2012; 36(5): 1142-47. doi: 10.1007/s00268-012-1504-5.
- Van-Santyoort HC, Besselink MG, Bakker OJ, Hofker HS, Boermeester MA, et al. A Step-up approach or open necrosectomy for necrotizing pancreatitis. New Engl J Med. 2010; 362: 1491-1502. doi: 10.1056/NEJMoa0908821
- Johnson CD, Abu-Hilal M. Persistent organ failure during the first week as a marker of fatal outcome in acute pancreatitis. Gut. 2004; 53(9): 1340- 1344. doi: 10.1136/gut.2004.039883
- Wu BU, Johannes RS, Sun X, Tabak Y, Conwell DL, Banks PA. The early prediction of mortality of acute pancreatitis: a large population-based study. Gut. 2008; 57(12): 1698-1703. doi: 10.1136/gut.2008.152702
- Al-Bahrani AZ, Darwish A, Hamza N, Benson J, Eddleston JM, Snider RH, et al. Gut barrier dysfunction in critically ill surgical patients with abdominal compartment syndrome. Pancreas. 2010; 39(7): 1064-1069. doi: 10.1097/ MPA.0b013e3181da8d51
- Banks PA, Bollen TL, Dervenis C, Gooszen HG, Johnson CD, Sarr MG, et al. Classification of acute pancreatitis 2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013; 62(1): 102-11. doi: 10.1136/gutjnl-2012-302779
- Sloss A, Kubler P. Prescribing in liver disease. Aust Prescr. 2009; 32(2): 32-5. doi: 10.18773/austprescr.2009.018
- Reintam A, Parm P, Kitus R, Starkopf J, Kern H. Gastrointestinal failure score in critically ill patients: a prospective observational study. Crit Care. 2008; 12(4): 90-95. doi: 10.1186/cc6958
- Hecker A, Hecker B, Hecker M, Riedel JG, Weigand MA, Padberg W. Acute abdominal compartment syndrome: current diagnostic and therapeutic options. Langenbecks Arch Surg. 2016; 401(1): 15-24. doi: 10.1007/s00423-015-1353-4
- Boyde TR, Rahmatullah M. Optimization of conditions for the colorimeteric determination of citrulline, using diacetyl monoxime. Anal Biochem. 1980; 107(2): 424-431. doi: 10.1016/0003-2697(80)90404-2
- Piton G, Manzon C, Cypriani B, Carbonnel F, Capellier G. Acute intestinal failure in critically ill patients: is plasma citrulline right market? Intensive Care Med. 2011; 37(6): 911-917. doi: 10.1007/s00134-011-2172-x
- Blasco Alonso J, Serrano Nieto J, Navas López VM, Barco Gálvez A, Vicioso I, Carazo Gallego B, et al. Plasma citrulline as a marker of loss of enterocitary mass in coeliac disease in childhood. Nutr Hosp. 2011; 26(4): 807-813. doi: 10.1590/S0212-16112011000400021
- Diamanti A, Panetta F, Gandullia P, Morini F, Noto C, Torre G, et al. Plasma citrulline as marker of bowel adaptation in children with short bowel syndrome. Langenbecks Arch Surg. 2011; 396(7): 1041-6. doi: 10.1007/ s00423-011-0813-8
- Lee EH, Ko JS, Seo JK. Correlations of plasma citrulline levels with clinical and endoscopic score and blood markers according to small bowel involvement in pediatric Crohn disease. J Pediatr Gastroenterol Nutr. 2013; 57(5): 570-575. doi: 10.1097/MPG.0b013e31829e264e


