Research Article
Structural, Thermal and Chemical Properties of Co-Cr Fe2O4 Nanocomposite Synthesized by Combustion Method
1Department of Physics, R.T.E.S. Society Science, Arts and Commerce College, Karnataka, India
2Department of physics, Mangalore University, Karnataka, India
3Academic Consultant, NAAC, Karnataka, India
*Corresponding author: Dr. Madhukumar Rajappa, Department of Physics, R.T.E.S. Society Science, Arts and Commerce College, Karnataka, India, E-mail: nwwton@gmail.com
Received: May 22, 2019 Accepted: May 27, 2019 Published: June 3, 2019
Citation: Harihar CA, Basavaraj HG, Guruswamy B, et al. Structural, Thermal and Chemical Properties of Co-Cr Fe2O4 Nanocomposite Synthesized by Combustion Method. Madridge J Nanotechnol Nanosci. 2019; 4(2): 156-159. doi: 10.18689/mjnn-1000131
Copyright: © 2019 The Author(s). This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Co-Cr Fe2O4 Nanocomposite were synthesized by solution combustion method and synthesized powder were characterized by X-Ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR) and Thermal Properties (DSC). The XRD results confirm the cubic spinel structure of the ferrites and crystalline size (D) found the range of 40-50 nm. The FTIR measurement between 400 – 4000 cm-1confirm the absorption bands in the Spectrum. Further, Thermogravimetric analysis and differential scanning calorimetry were used to investigate the phase transition and melting point of the prepared samples respectively. Agglomeration of particles was observed in the scanning electron microscopy (SEM) images. The results underline the effect of preparation conditions on the morphology, crystallite size, and thermal properties of nano ferrites.
Keywords: Nanocomposite, XRD, FTIR, DSC, SEM.
Introduction
In the last few years, ferrites have been the emerging focus of recent scientific research and technological there has been growing research on the investigations of ferrite nanostructures. During the past decades, Ferrites have proved to be good in microwave applications because of their low cost, high resistivity and low eddy current losses [1] microwave absorption materials have received remarkable attention due to their unique electronic and magnetic properties and their potential application in various fields, especially in electromagnetic interference shielding and radar systems. Nanocrystalline ferrite materials are attracting an increasing interest nowadays. Owing to the small characteristic size of their nanostructure, they exhibit novel properties which differ from those of materials with micron-sized features. Recently, Nickel nanoferrite, an important member of ferrite family, has attracted major research interest due to its applications in technological devices such as circulators, isolators, gyrators, phase shifters, filters, and switches and substrates for microwave integrated circuits [2,3]. Various works present the preparation of ferrites using a conventional ceramic powder preparation process, which involves a solid state reaction. This technique has disadvantages, such as: formation of strongly bonded agglomerates, non-homogeneities, such as: undesirable phases, abnormal grain growth, poor reproducibility and imprecise control of the cation stoichiometry and ratios. The combustion synthesis technique has proved to be a novel, extremely facile, time-saving and energy-efficient route for the synthesis of ultra-fine powders. The combustion method presents some advantages compared to other methods: reagents are very simple compounds, special equipment is not required and dopants can be easily introduced into the final product. In the present work, Nickel nanoferrites were prepared by solution combustion method and dielectric & a.c. conductivity studies on the as prepared Co-Cr Fe2O4 nanoparticles have been undertaken over a wide frequency range (100Hz-5MHz) at room temperature [4].
Experimental
The Co-Cr Fe2O4 nanoferrite powder has been prepared by solution combustion method using stoichiometric composition of Co-Cr nitrate as oxidizer and urea as a fuel. The aqueous solution containing redox mixture was taken in a Pyrex dish and heated in a muffle furnace maintained at 500 ± 10°C. The mixture finally yields porous and voluminous powder (Figure 1).
The X-ray diffractograms of the synthesized samples were recorded using Panalytical X-Pert Pro MPD instrument. The samples were scanned in the 2θ range of 10-70°, with a scanning speed and step size of 5°/min and 0.02°, respectively. Fourier transform infrared (FTIR) spectra of the samples were recorded in transmission mode using Thermo Nicolet, Avatar 370, FTIR spectrophotometer having a resolution 4 cm-1 in the wave number range 400-4000 cm-1. Samples were mixed with KBr powder for FTIR measurements. Background correction was made using a blank KBr pellet as a reference.
The morphology of the synthesized samples were analyzed using Field emission scanning electron microscopy (FE-SEM) attached with Energy Dispersive X-ray (EDX) analysis (ZEISS). This microscope is equipped with a field emission gun, operating at an accelerating voltage variable from 0.5 to 30 kV, with a resolution of 2 nm.
Thermo gravimetric analysis (TGA) of the sample was carried out by Perkin Elmer Thermal Analysis system with nitrogen as flushing gas. The temperature range scanned was 25°C - 700°C at a predetermined rate of 20°C/min.

Results and Discussions
X-ray Diffraction (XRD) analysis
Figure 2 shows the X-ray diffraction pattern of Co-SrFe2O4 powder sample. The grain sizes of the sample were evaluated by measuring the FWHM. The XRD patterns of the Co-Cr Fe2O4 Nanocomposite of crystalline phases were identified by comparison with reference data from the ICSD card No. 22-1086. Figure 2 shows the X-ray diffraction pattern of Co-Cr Fe2O4 nanocomposites. Analysis of X-ray diffraction pattern revealed the formation of single spinel phase. The average crystallite size of the Co-Cr Fe2O4 Nanocomposite determined by the Debye-Scherrer formula, where D is the crystallite size, k = 0.9 is a correction factor to account for the particle shapes, b is the full width at a half maximum of the most intense diffraction peak (311) plane, l is the wavelength of a Cu Ka radiation (1.5418 Å) and u is the Bragg angle.
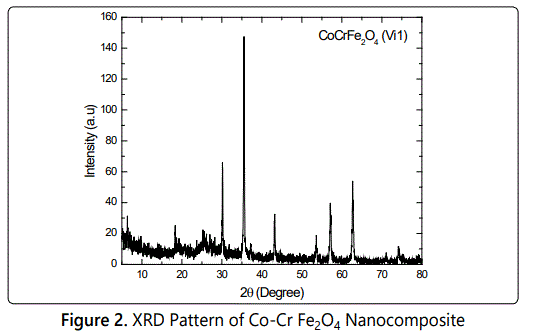
The average crystallite size of the prepared sample is around 46 nm. Observed peak at 2θ = 35.630 C indicates the semi crystalline nature of nanoferrites present within the sample [5]. They all show the reflection planes (111), (220), (311), (222), (400), (422), (511) and (440), corresponding to a crystalline cubic, spinel-type phase. They provide clear evidence of a series of solid between Co-Cr Fe2O4 Nanocomposite. The patterns show a slight shift in peak position towards lower d-spacings [6].
Scanning Electron Morphology (SEM)
The microstructural morphology observations have been performed on Co-Cr Fe2O4 nanocomposites using Scanning Electron Microscope (SEM). Figure 3 shows the SEM image of Co-Cr Fe2O4 Nanocomposite. The SEM image displays the distribution of the Co-Cr Fe2O4 nanoferrities particles. Co-Cr Fe2O4 Nanocomposite is flaky and appears as aggregates of irregular shapes with diameter ranging from 35-65 nm [7].

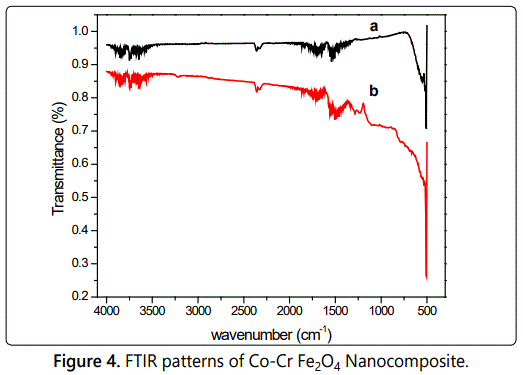
The FTIR spectra of Co-Cr Fe2O4 Nanocomposite shown in Figure 4.The presence of peaks at 1556 cm-1 and 1482 cm-1 (Figure 4) confirms the polymerization of aniline [8]. Characteristic peaks of polyaniline are found at 1679 cm-1, 1556 cm-1 and 1489 cm-1. The peaks at 1556 cm-1 and 1493 cm-1 corresponds to C=C stretching deformation of quinonoid and benzenoid units respectively. The peak observed at 1284 cm-1 attributes C-N stretching of secondary amine in polymer main chain [9]. The bands at 1400–1625 cm-1 and 1128 cm-1 of the composites (Figure 4) indicates the coupling effect of ferrite and polyaniline [10].
Thermogravimetric analysis (TGA)
TGA curve demonstrates a weight loss of about 14% which may be due to the decomposition and oxidation of organic substances. Moreover, the formation of monophasic ferrite exhibits the drastic weight loss of about 31% at 580 °C. It can also be observed from the curve that there is no considerable weight loss thereafter 600 °C which indicates that the formation of spinel phase at this temperature.

Figure 5 shows the TGA curves obtained for as on prepared samples with chemical composition of Co-Cr Fe2O4 and involves only two transitions physical and chemical during decomposition. Usually decomposition starts to occur at about 100 °C with loss of water of crystallization, combustion, reduction of metal oxides and physical transition like vaporization and evaporation. Ferrites usually have higher melt temperature round 900 °C [11-13]. For the composition of Co-Cr Fe2O4, in the first region, starting from room temperature up to 325 °C, the weight loss could be attributed to the elimination of adsorbed water. The weight change was not significant and the sample was thermally stable. In the second region from 390.25 to 525.23 °C, the film experienced a great weight loss about 50% of the sample decomposed into volatiles. Another strong (the third region) weight loss of about 85% in the range 650–830 °C. Co-Cr Fe2O4 composites also exhibited three distinct regions with small variation in the decomposition temperature.
Conclusion
The Co-Cr Fe2O4 Nanocomposite were synthesized by solution combustion technique. As-prepared samples were examined by using XRD, FT-IR and FE-SEM analysis techniques. Thermal stability of the samples was analyzed using TGA. The average crystallite size of the sample is found to be 64.11 nm. Analysis of the X-ray diffraction pattern revealed the nanocrystalline nature of the as prepared sample. The SEM image of the synthesized Co–Srnanoferrites,which confirms the nanoscale range of the prepared Co-Cr Fe2O4 Nanocomposite.
Acknowledgments
One of the Authors are grateful to MIT Manipal for SEM and UPE, University of Mysore for XRD experiments
References
- Chand J, Verma S, Kumar P, Singh M. Structural, Electric and Dielectric Properties of MgFe2O4 Ferrite Processed by Solid State Reaction Technique. Inter. J. of theor. Appl. Sci. 2011; 3(2): 8-9.
- Patil KC, Hegde MS, Rattan T, Aruna ST. Chemistry of Nanocrystalline Oxide Materials: Combustion Synthesis, Properties and Applications. World Scientific Publishing. 2008; 1-364.
- Kulkarni SK. Nanotechnology: principles and practices. Capital publishing company. 2009.
- Soibam I, Phanjoubam S, Prakash C. Magnetic and Mössbauer studies of Ni substituted Li–Zn ferrite. J. Magn. Magn. Mater. 2009; 321(18): 2779-2782.
- Kondawar SB, Arti, Nandapure AI, Nandapure BI. Nanocrystalline Nickel Ferrite Reinforced Conducting Polyaniline Nanocomposites. Adv. Mat. Lett. 2014; 5(6): 339-344. doi: 10.5185/amlett.2014.amwc.1035
- Madhu BJ, Angadi VJ, Mallikarjuna H, Manjunatha SO, Shruthi B, Kumar RM. Dielectric Behavior and A. C. Conductivity Studies on Nickel Nanoferrites Synthesized by Solution Combustion Method. Adv. Mater. Res. 2012; 584: 299-302. doi: 10.4028/www.scientific.net/AMR.584.299
- Khairy M. Synthesis, characterization, magnetic and electrical properties of polyaniline/NiFe2O4 nanocomposite. Synthetic Metals. 2014; 189: 34-41. doi: 10.1016/j.synthmet.2013.12.022
- Jiang J, Li L, Xu F. Polyaniline–LiNi ferrite core–shell composite: Preparation, characterization and properties. Mater Sci Eng A. 2007; 456(1): 300-304. doi: 10.1016/j.msea.2006.11.143
- Madhu BJ, Gurusiddesh M, Kiran T, Shruthi B, Jayanna HS. Structural, dielectric, ac conductivity and electromagnetic shielding properties of polyaniline/Ni0.5Zn0.5Fe2O4 composites. J Materials Science: Materials in Electronics. 2016; 27(8): 7760-7766. doi: 10.1007/s10854-016-4764-2
- Raghavender AT, Jadhav KM. Dielectric properties of Al-substituted Co ferrite nanoparticles. Bull. Mater. Sci. 2009; 32(6): 575-578. doi: 10.1007/s12034-009-0087-8
- Hsu JY, Ko WS, Shen HD, Chen CJ. Low temperature fired NiCuZn ferrite. IEEE Trans Magn. 1994; 30(6): 4875-4877. doi: 10.1109/20.334251
- Jean JH, Lee CH, Kou WS. Effects of Lead(II) Oxide on Processing and Properties of Low-Temperature-Cofirable Ni-Cu-Zn Ferrite. J. Amer. Ceram Soc. 1999; 82(2): 343-350. doi: 10.1111/j.1551-2916.1999.tb20068.x


