Research Article
Resistive Switching behaviour in Oxide Ion Conductor Na0.5Bi0.5Ti0.98Mg0.02O3-δ Thin Films
State Key Laboratory of MMCs, School of Materials Science and Engineering, Shanghai Jiaotong University, China
*Corresponding author: Yiping Guo, State Key Laboratory of MMCs, School of Materials Science and Engineering, Shanghai Jiaotong University, China, E-mail: ypguo@sjtu.edu.cn
Received: April 7, 2017 Accepted: April 13, 2017 Published: April 18, 2017
Citation: Guan L, Guo Y, Wu F, Duan H, Li H, Liu H. Resistive Switching behaviour in Oxide Ion Conductor Na0.5Bi0.5Ti0.98Mg0.02O3-δ Thin Films. Madridge J Nanotechnol Nanosci. 2017; 2(1): 52-57. doi: 10.18689/mjnn-1000110
Copyright: © 2017 The Author(s). This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Sol-gel derived perovskite oxide ion conductor thin films with a nominal composition of Na0.5Bi0.5Ti0.98Mg0.02O3-δ (NBTM) were fabricated on the In2O3-doped SnO2 (ITO) glass substrates. Bipolar resistive switching requiring no “electroforming” process and exhibiting ultrahigh resistance ON/OFF (RON/OFF) ratio (>104) were observed in the ITO/NBTM/Au capacitor. A model concerning the oxygen ion migration induced ionic conduction variation was proposed for the observed resistive switching (RS) effect, evidencing by a voltage-polarity dependent rectification switching behaviour, and the I-V behaviour in high purity argon atmosphere. Our model demonstrates that the migration of oxygen ions can reverse the resistance just by modulating the oxygen ionic conduction, which is different from those previously proposed ones relating to oxygen-ions, e.g. the chemical redox reactions of the oxides or active electrodes, the composition changing of the RS materials, or the Schottky-barrier height modulating of the metal/oxide interfaces.
Keywords: Na0.5Bi0.5TiO3; Oxide ion conductor; Resistive switching; Oxygen ion conduction.
Introduction
Perovskite sodium bismuth titanate Na0.5Bi0.5TiO3 (NBT) is a well-known lead-free piezoelectric material, which has potentials to replace the Pb(Zr1-xTix)O3-based piezoelectrics for the development of environmentally-friendly ceramic-based capacitors. The electrical properties of perovskite metal oxides are susceptible to doping by a variety of defects and impurities. Many are intrinsically “self-doped” by native interstitials or vacancy point defects [1]. Li et al. [2] have recently reported that the doped-NBT materials are not only good piezoelectrics but also a new family of oxide ion conductors, in which the oxygen ionic conduction rather than the electronic conduction (electrons or holes) is dominant. They have demonstrated that the NBT lattice is a good structural host for oxygen ionic conductivity because of the highly polarized Bi3+ as well as the weak Bi-O bonds. They have also pointed out that the nominal composition tuning of NBT will lead to the conductivity variation, eg. Mg-doping in the Ti-site of NBT increases the oxygen ionic conductivity by enlarging the concentration of oxygen vacancies. Their work has provided a concept of preparing electronic devices ranging from dielectricbased to conductor-based devices with the NBT system by adjusting the nominal composition of NBT.
Resistive switching (RS) is a large, reversible and nonvolatile change in the resistance after the applications of voltage or current pulses [3]. A typical RS memory unit has a capacitor-like structure composed of an insulating or semiconducting material sandwiched between two metal electrodes [4]. Among the materials exhibiting RS phenomena, oxides have been studied intensively, due to their potentials for using as new types nonvolatile memories. Oxides reported to exhibit RS effects include complex perovskite oxides [5,6], binary transition metal oxides [7-10] and some main oxides [11] as well. Up to now, the details for the RS effects in oxides have not been well understood, various theoretical models have been suggested. Among them, models concerning the oxygen vacancies or oxygen ions have been the mostly proposed ones [12]. These models mainly concern the effects brought by the migration of oxygen ions, which indirectly influence the conductivity of the RS cells. The effects include: chemical redox reactions at the oxide/electrode interface, in which the involved electrodes are often electrochemically active and can be easily oxidized like Al, Ti, Ir, etc. [13-17]; chemical redox reactions at the bilayer oxide films interface with different initial oxygen ion or oxygen vacancy distributions in each film [18]; stoichiometric changes causing the formation and rupture of conductive filaments [10,19]; stoichiometric changes introducing different compositions of the switching materials with different conductivities to dominate the reversible resistance states [20]; valence changes of the switching materials changing the local electronic conductivity [21]; Mott-transition of the switching materials relating to the electronic effect [22]; oxygen deficiency defects introduced at the metal (often Pt)/oxides interface changing the Schottky-barrier height at the interface [15]; formation of the interface oxide layer at the electrode/oxide interface, modulating the Schottky barrier height [23]; etc. Among these oxides with RS phenomena, some are either highly intrinsic insulating which needs an “electroforming” process to change the resistance behaviours [10,13,20,24,25] or semiconducting (p-type or n-type) relying mainly on electronic conduction rather than ionic conduction.
To the best of our knowledge, few papers have reported the RS effects of the newly reported doped-NBT oxide ion conductors (electrolytes), and many of the ever reported RS effects concerning the electrolytes have been suggested to be related to the chemical redox reactions of the metal electrodes like Ag, Cu, Ni, etc. [26-32]. In this paper, we reported a RS effect in the sol-gel derived Na0.5Bi0.5Ti0.98Mg0.02O3-δ (NBTM) oxide ion conductor thin films. It is proposed that the migration of oxygen ions can reverse the resistance just by modulating the oxygen ionic conduction, which is different from those previously proposed ones relating to oxygen-ions, e.g. the chemical redox reactions of the oxides or active electrodes, the composition changing of the RS materials, or the Schottky-barrier height modulating of the metal/oxide interfaces.
Experimental
The NBTM polycrystalline thin films with thickness of 150 nm were deposited on the commercial In2O3-doped SnO2 (ITO) glass substrates by a two-step chemical solution deposition (CSD) method as reported in our previous work [33]. The 0.3 mol/L precursor solutions were synthesized starting from tetrabutyltitanate (Ti [O(CH2)3CH3]4), sodium nitrate (NaNO3), bismuth nitrate (Bi(NO3)3·5H2O), and magnesium nitrate (Mg(NO3)2·6H2O). Acetylacetone (CH3COCH2COCH3) was adopted as ligand. 2-methoxyethanol (HOCH2CH2OCH3) and acetic acid (CH3COOH) were chosen as co-solvents. The precursor films were spin coated on the ITO glass substrates at 1000 rpm for 10 s and then 3000 rpm for 30 s. After coating each layer, the thin films were dried at 150 °C for 3 min, pre-fired at 350 °C for 5 min in air, and then rapidly thermal annealed at 650 °C for 5 min in oxygen atmosphere. These steps were repeated eight times.
The crystallographic structures of the films were characterized by X-ray diffraction (XRD) analysis (Rigaku D/max-2550/PC with Cu Kα radiation) with a scan speed of 5 °/min. The Au top electrodes were deposited on NBTM films by ion-sputtering though rectangular masks (1 mm × 0.5 mm) to fabricate the ITO/NBTM/Au capacitors for electrical measurements. In order to determine the contribution of oxygen for the RS effect, the current–voltage (I–V) characteristics of the NBTM thin film RS memory devices was measured using a Keithley 2400 source meter in air or in glove box with high purity argon, respectively. The piezoelectric force microscopy (PFM) (Asylum Research/Cypher) was used to study the domain back-switching behaviours of the NBTM thin films. The films were poled through the PFM tip by scanning the film surface with a positive +9 V voltage in a 1μm×1μm area and a negative -9 V one in a 3μm × 3μm area. The piezoelectric phase image was carried out by applying an ac voltage (frequency 2.44 Hz, amplitude 2 V) to the PFM tip in a square area of 5μm × 5μm.
Results and Discussions
Figure 1 shows the XRD patterns of the as-prepared NBTM thin films grown on the ITO glass substrates. No other peaks are detected except those coming from the substrates for In2O3, SnO2, and the perovskite. All the peaks can be indexed according to the standard powder diffraction data of Na0.5Bi0.5TiO3, indicating that the Na0.5Bi0.5Ti0.98Mg0.02O3-δ thin films were polycrystalline with pure perovskite phase.
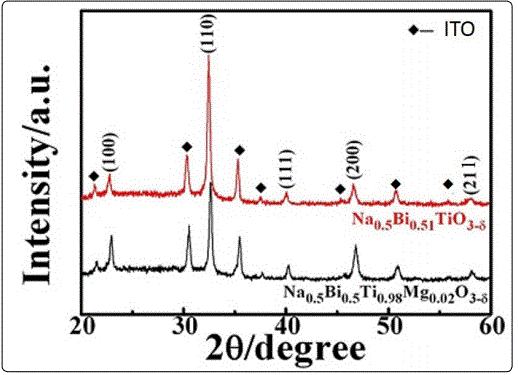
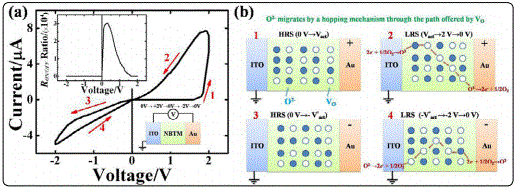
Figure 2 shows the I-V curves of the ITO/NBTM/Au capacitor under the bias voltage sweep of 0 V → +2 V → 0 V → -2 V → 0 V. Herein, the positive forward bias is defined when a positive bias voltage applied to the Au top electrode. A typical I-V hysteresis of the ITO/NBTM/Au capacitor was observed without any “electroforming” process, indicating a large bipolar resistive switching with bistable resistance states. The RON/OFF ratio (defined as the ratio of ON state current divided by OFF state one at a voltage) is larger than four orders of magnitude in the positive diode, and the maximum value of the RON/OFF ratio is 30,159 at the reading voltage of +0.3 V. The RS behaviours are repeatable and each state persists in the low voltage regime. When discussing the origin of the RS effect, we firstly rule out the possibility that the chemical redox reactions of the electrodes are responsible for the RS effect, because no electrochemically active electrodes were adopted in our work. Moreover, the interface-type Schottky-like band modification induced by the electro migration of oxygen vacancies can also be ruled out, because this model cannot explain the voltage-polarity dependent rectification switching of the ITO/NBTM/Au capacitor, which will be discussed later. The plausible explanation of the I-V hysteresis is the resistance variation resulted from the electromigration of oxygen ions, as Li et al have suggested that a predominance of oxygen ionic conduction rather than the electronic conduction exhibits in the Mg-doped or Bi-deficient NBT [2]. The migration of oxygen ions brings in ionic conduction variation of the NBTM film. In polycrystalline thin films, there are a lot of grain boundary defects, and the resistivity of the grain boundaries is much higher than that of the grains interiors [34]. The fresh NBTM sample is most likely to be in the high resistance state (HRS) because of the large grain boundary resistance [35]. Acceptor-doping of Mg increases the oxygen ion conductivity by increasing the concentration of oxygen vacancies, which provides the diffusion paths for the migration of oxygen ions in the lattices. Oxygen ions are suggested to be highly mobile under an external electric field in many metal oxides [2,36]. When applying a voltage sweep from 0 V to +2 V on the Au electrode, the current is at first very small until a certain high positive bias voltage (defined here as the activating voltage) is applied, which is large enough to activate the oxygen ions to get rid of the bound of the lattices. The oxygen ions will thus migrate towards the Au electrode by a hopping mechanism, where they release two electrons to the electrode to evolve into the oxygen gas. At the same time, the absence of oxygen ions introduces oxygen vacancy defects, equivalent to the redistribution of oxygen vacancies near the ITO electrode. The oxygen vacancies then absorb the oxygen gas from the air, taking electrons from the ITO electrode and being reduced to oxygen ions [10,21]. The migration of oxygen ions accompanies with the large ionic conductivity, switching the sample into the low resistance state (LRS). The samples retains in LRS with the voltage sweeping from +2 V to 0 V. This also applies to the condition with the negative voltage sweeping range of 0 V → -2 V→ 0 V. That is to say, with the negative bias voltage sweep from 0 V to -2 V, the sample is in HRS at first, and when the reverse voltage is large enough, the sample is switched into LRS. The sample retains in LRS under the negative bias voltage sweep from -2 V to 0 V. The high asymmetry of I-V hysteresis (ON state current in the positive diode is much larger than the one in the negative diode) may be attributed to the asymmetric distribution of oxygen vacancies along the ITO/NBTM/Au packing direction, because the oxygen vacancy density near the top surface of the thin film is much higher than the equilibrium concentration [21]. And the high oxygen vacancy concentration at the Au/NBTM interface is beneficial for the oxygen ion diffusion in the NBTM oxide ion conductor [2]. The asymmetric ITO/NBTM/Au structure is probably responsible for the unequal activating voltages in the positive and negative range.
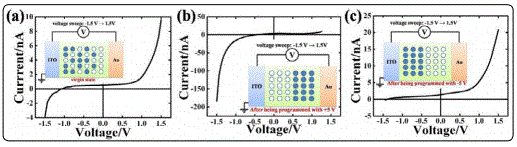
To elucidate the models proposed for explaining the RS effects in NBTM films, it is necessary to clarify the influence of the electromigration of oxygen ions to the resistance variations of the ITO/NBTM/Au capacitors. An effective way to change the oxygen ion distribution in the film is to apply a high bias voltage on the capacitor [37], which is defined here as the “programming” procedure. By measuring the I-V curves of the “programmed” samples, the impact of the redistribution of oxygen ions under bias voltage on the conduction behaviours can be observed. Figure 3 is the I-V curves with bias voltage sweeping from -1.5 V to +1.5 V for the ITO/NBTM/Au capacitors at the virgin and the “programmed” state. The “programming” procedure was operated by applying a +5 V (-5 V) bias voltage on the capacitors for 1min. Reversible and distinct switchable diode effects were observed for the samples “programmed” by bias voltage with reverse polarities. The rectification ratio (defined as the ratio of the forward current divided by the reverse on at voltages with the same amplitude) of the +5 V (-5 V) “programmed” samples is 5 (128) at 1.4 V. The virgin NBTM sample exhibits asymmetric I-V curves, indicating an asymmetric potential distribution resulted from the uneven oxygen vacancy distribution [21]. When “programming” the sample with +5 V bias voltage for 1 min, oxygen ions will be fully activated and accumulate near the Au electrode while oxygen vacancies will pile up near the ITO electrode (The schematic diagram shown in the inset of Figure 3(b) represents the ideal state of the distribution of oxygen ions and oxygen vacancies after being “programmed” with +5 V voltage.). Under the negative sweeping voltage, oxygen ions will migrate towards the oxygen vacancies sufficient ITO electrode, where they release electrons and be oxidized to oxygen gas, whilst oxygen vacancies migrate towards the Au electrode where they take the electrons, reducing the absorbed oxygen gas to oxygen ions, the migration of oxygen ions enhances the ionic conduction, bringing in high conductivity; while under the positive sweeping voltage, oxygen ions are difficult to move due to the oxygen vacancy deficiency near the Au electrode, hindering their oxidation and resulting in low conductivity. Similarly, when “programming” the sample with -5 V bias voltage (The schematic diagram shown in the inset of Figure 3(c) represents the ideal state of the distribution of oxygen ions and oxygen vacancies after being “programmed” with -5 V voltage.), low conductivity will rise in the negative sweeping voltage range while high conductivity in the positive sweeping voltage range.
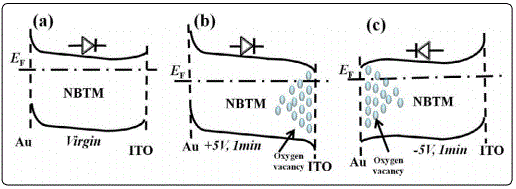
The authors notice that a similar voltage-polarity dependent reproducible rectification diode switching phenomenon has been reported in the Pt/TiOx/Pt trilayer capacitors [38]. However, the switching materials as TiOx and NBTM are two different materials in nature. The former is often considered as an n-type oxide relying on the electronic conduction due to the naturally produced oxygen vacancies acting as donors [38], while the latter has been recently reported to be an oxide ion conductor with a predominance of oxygen ion conduction [2]. Besides, contradictions would arise if we consider the NBTM as an n-type oxide concerning the presence of oxygen vacancies acting as donors, to explain the observed voltage-polarity dependent reproducible rectification switching phenomenon. Firstly, for the hypothetic n-type NBTM thin film at the virgin state, the asymmetry leakage J-V curve of the ITO/NBTM/Au capacitor might indicate the formation of asymmetric Schottky barrier contacts at the ITO/NBTM interface and the Au/NBTM interface due to work functions variations, and the Schottky barrier height of the Au/NBTM contact is higher than that of the ITO/NBTM contact, resulting in a forward diode (Figure 4(a)). As oxygen vacancies act as effective donors in n-type oxides, the reduction (increase) in the concentration of oxygen vacancies may widen (narrow) the depletion region and increase (decrease) the contact resistance [4]. Thus, when a positive bias voltage is applied on the capacitor, positively charged oxygen vacancies (or) will pile up near the ITO/NBTM interface, leading to an enrichment of or near the ITO/NBTM interface and an depletion of or near the Au/NBTM interface, which therefore decreases the barrier height of the ITO/NBTM contact while increases the barrier height of the Au/NBTM contact and results in a band bending as shown in Figure 4(b). Similarly, when a negative bias voltage is applied, a resultant band bending shown in Figure 4(c) would induce a reverse diode. Both of which are contradictory to what we have observed in our work.
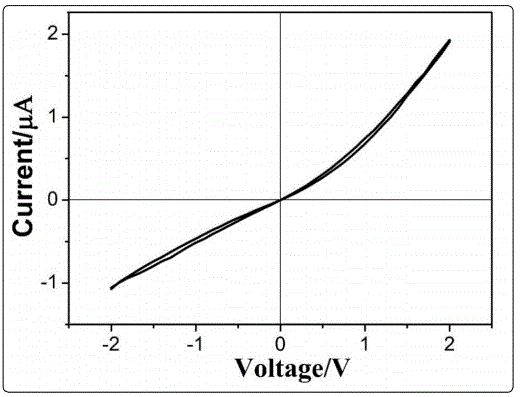
Herein, we also reported a very simple method to disentangle the contribution of electron or ion to the RS effect. We measured the I–V curve with a Keithley 2400 source meter in a glove box with high purity argon. Fig.5 shows the I-V curve for the ITO/NBTM/Au capacitor under the bias voltage sweep of 0 V → +2 V → 0 V → -2 V → 0 V. The RS effect disappears and the conductivity significantly decreases. This discovery confirmed that the oxygen gas is necessary to get the RS effect in oxygen ion conductor NBTM.
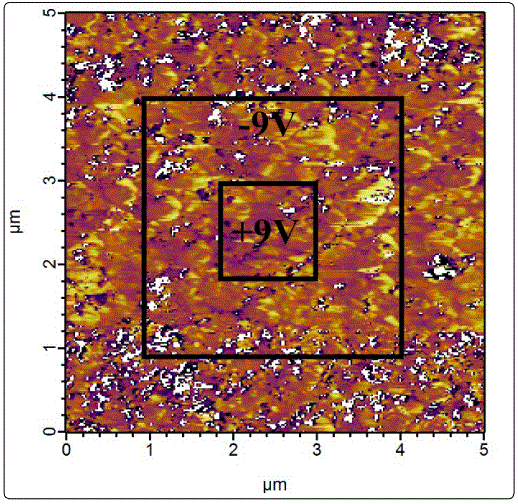
To figure out whether or not the voltage-polarity dependent switchable diode effect is triggered by the ferroelectric reversal, the piezoelectric force microscopy (PFM) was used to measure the domain back-switching behaviours of the NBTM thin film samples. Results suggest that the switchable diode effect of the samples “programmed” with ±5 V voltage barely has anything to do with the ferroelectric reversal as the domain back-switching was not observed for the samples polarized with ±9 V (Figure 6). However, it is possible that ferroelectric reversal plays a part on the switchable diode effect of the samples “trained” with higher voltages, eg. ±15 V (results are not shown here), which has been observed in both polycrystalline and epitaxial BiFeO3 thin films [37,39]. It is a pity that the PFM is not capable of elucidating the impacts of ferroelectric reversal on the switchable diode effects of the ±15 V “programmed” samples due to the instrument condition with a ±10 V maximum poling voltage. Such poling voltage of ±10 V is not large enough to reverse the domain phase of NBTM because of the large coercive field, resulted from the domain pinning effects of the large concentration of the oxygen vacancy defects [40].
Conclusion
In summary, switchable diode and resistive switching effects were observed in the Mg-doped NBT oxide ion conductor thin film based capacitors. The redistribution of oxygen ions under the bias voltage is thought to play a key role in the switchable diode and resistive switching effects. Our work launches a new type of potential candidates— the perovskite doped-NBT oxide ion conductors for the RS memory cells and broadens the categories of oxide materials exhibiting RS effects.
Acknowledgements
This work is supported by the National Natural Science Foundation of China (NO. 11474199 and NO.51332009). The authors acknowledge Prof. Xiang of the Key Laboratory of Polar Materials and Devices, Ministry of Education, East China Normal University for the assistance of PFM measurements.
References
- Yang JJ, Pickett MD, Li X, Ohlberg DA, Stewart DR, Williams RS. Memristive Switching Mechanism for Metal/Oxide/Metal Nanodevices. Nat. Nanotechnol. 2008; 3: 429-33. doi: 10.1038/nnano.2008.160
- Li M, Pietrowski MJ, De Souza RA, et al. A Family of Oxide Ion Conductors Based on the Ferroelectric Perovskite Na0.5Bi0.5TiO3. Nat. Mater. 2014; 13: 31-35. doi: 10.1038/nmat3782
- Rozenberg MJ, Sánchez MJ, Weht R, Acha C, Gomez-Marlasca F, Levy P. Mechanism for Bipolar Resistive Switching in Transition-metal Oxides. Phys. Rev. B. 2010; 81:115101. doi: 10.1103/PhysRevB.81.115101
- Sawa A. Resistive Switching in Transition Metal Oxides. Mater. Today. 2008; 11: 28-36. doi: 10.1016/S1369-7021(08)70119-6
- Tsubouchi K, Ohkubo I, Kumigashira H, et al. High-throughput Characterization of Metal Electrode Performance for Electric-field-induced Resistance Switching in Metal/Pr0.7Ca0.3MnO3/Metal Structures. Adv. Mater. 2007; 19(13): 1711-13. doi: 10.1002/adma.200601957
- Hong S, Choi T, Jeon JH, et al. Large Resistive Switching in Ferroelectric BiFeO3 Nano-island Based Switchable Diodes. Adv. Mater. 2013; 25: 2339-343. doi: 10.1002/adma.201204839
- Yoon KJ, Song SJ, Seok JY, et al. Ionic Bipolar Resistive Switching Modes Determined by the Preceding Unipolar Resistive Switching Reset Behavior in Pt/TiO2/Pt. Nanotechnology. 2013; 24(14): 145201. doi: 10.1088/0957-4484/24/14/145201
- Do YH, Kwak JS, Hong JP, Jung K, Im H. Al Electrode Dependent Transition to Bipolar Resistive Switching Characteristics in Pure TiO2 Films. J. Appl. Phys. 2008; 104: 114512. doi: 10.1063/1.3032896
- Son JY, Shin YH. Direct Observation of Conducting Filaments on Resistive Switching of NiO Thin Films. Appl. Phys. Lett. 2008; 92: 222106. doi: 10.1063/1.2931087
- Chen JY, Hsin CL, Huang CW, et al. Dynamic Evolution of Conducting Nanofilament in Resistive Switching Memories. Nano Lett. 2013; 13(8): 3671-77. doi: 10.1021/nl4015638
- Do YH, Kwak JS, Bae YC, Jung K, Im H, Hong JP. Oxygen Ion Drifted Bipolar Resistive Switching Behaviors in TiO2-Al Electrode Interfaces. Thin Solid Films. 2010; 518(15): 4408-411. doi: 10.1016/j.tsf.2010.01.016
- Kim KM, Choi BJ, Hwang CS. Localized Switching Mechanism in Resistive Switching of Atomic-Layer-Deposited TiO2 Thin Films. Appl. Phys. Lett. 2007; 90: 242906. doi: 10.1063/1.2748312
- Huang JJ, Kuo CW, Chang WC, Hou TH. Transition of Stable Rectification to Resistive-Switching in Ti/TiO2/Pt Oxide Diode. Appl. Phys. Lett. 2010; 96(26): 262901. doi: 10.1063/1.3457866
- Senthilkumar V, Kathalingam A, Kannan V, Senthil K, Rhee JK. Reproducible Resistive Switching in Hydrothermal Processed TiO2Nanorod Film for Non-Volatile Memory Applications. Sensor Actuat. A: Phys. 2013; 194: 135-39. doi: 10.1016/j.sna.2013.02.009
- Lin CY, Wu CY, Wu, CY, et al. Effect of Top Electrode Material on Resistive Switching Properties of ZrO2 Film Memory Devices. IEEE Electron Device Letters. 2007; 28(5): 366-68. doi: 10.1109/LED.2007.894652
- Bae YC, Lee AR, Lee JB, et al. Oxygen Ion Drift-Induced Complementary Resistive Switching in Homo TiOx/TiOy/TiOx and Hetero TiOx/TiON/TiOx Triple Multilayer Frameworks. Adv. Funct.Mater. 2012; 22(4): 709-16. doi: 10.1002/adfm.201102362
- Yoshida C, Kinoshita K, Yamasaki T, Sugiyama Y. Direct Observation of Oxygen Movement During Resistance Switching in NiO/Pt Film. Appl. Phys. Lett.2008; 93: 042106. doi: 10.1063/1.2966141
- Yoshida C, Tsunoda K, Noshiro H, Sugiyama Y. High Speed Resistive Switching in Pt/TiO2/TiN Film for Nonvolatile Memory Application. Appl. Phys. Lett. 2007; 91: 223510. doi: 10.1063/1.2818691
- Gao P, Wang Z, Fu W, et al. In Situ TEM Studies of Oxygen Vacancy Migration for Electrically Induced Resistance Change Effect in Cerium Oxides. Micron. 2010; 41(4): 301-05. doi: 10.1016/j.micron.2009.11.010
- Fujimoto M, Koyama H, Konagai M, et al. TiO2 Anatase Nanolayer on TiN Thin Film Exhibiting High-Speed Bipolar Resistive Switching. N.Appl. Phys. Lett. 2006; 89(22): 223509. doi: 10.1063/1.2397006
- Haman M, Dong R,Choi HJ, et al. Effect of Ruthenium Oxide Electrode on the Resistive Switching of Nb-doped Strontium Titanate. Appl. Phys. Lett. 2008; 93: 052908. doi: 10.1063/1.2969052
- Jeong DS, Schroeder H, Waser R. Coexistence of Bipolar and Unipolar Resistive Switching Behaviors in a Pt/TiO2/Pt Stack. Electrochem. SolidState lett. 2007; 10(8): G51-G53. doi: 10.1149/1.2742989
- Tsunoda K, Fukuzumi Y, Jameson JR, Wang Z, Griffin PB, Nishi Y. Bipolar Resistive Switching in Polycrystalline TiO2 Films. Appl. Phys. Lett. 2007; 90: 113501. doi: 10.1063/1.2712777
- Sakamoto T, Sunamura H, Kawaura H, Hasegawa T, Nakayama T, Aono M. Nanometer-Scale Switches Using Copper Sulfide. Appl. Phys. Lett. 2003; 82: 3032. doi: 10.1063/1.1572964
- Choi SJ, Park GS, Kim KH, et al. In Situ Observation of Voltage-induced Multilevel Resistive Switching in Solid Electrolyte Memory. Adv. Mater. 2011; 23(29): 3272-77. doi: 10.1002/adma.201100507
- Sun J, Liu Q, Xie H, et al. In Situ Observation of Nickel as an Oxidizable Electrode Material for the Solid-Electrolyte-Based Resistive Random Access Memory. Appl. Phys. Lett. 2013; 102(5): 053502. doi: 10.1063/1.4790837
- Wang Z, Griffin PB, McVittie J, Wong S, McIntyre PC, Nishi Y. Resistive Switching Mechanism in ZnxCd1-xS Nonvolatile Memory Devices. Electron Dev. Lett.; IEEE. 2007; 28(1): 14-16. doi: 10.1109/LED.2006.887640
- Xu Z, Bando Y, Wang W, Bai X, Golberg D. Real-time in Situ HRTEMResolved Resistance Switching of Ag2S Nanoscale Ionic Conductor. ACS Nano. 2010; 4(5): 2515-22. doi: 10.1021/nn100483a
- Guo B, Guo Y, Qin W, Liu H, Zhang D. Optical Properties of BiFeO3-(Na0.5Bi0.5)TiO3 Thin Films Deposited on Glass Substrates by Chemical Solution Deposition. Mater. Lett. 2012; 71: 60-62. doi: 10.1016/j.matlet.2011.12.042
- Yin K, Li M, Liu Y, et al. Resistance Switching in Polycrystalline BiFeO3 Thin Films. Appl. Phys. Lett. 2010; 97: 042101. doi: 10.1063/1.3467838
- Waser R, Aono M. Nanoionics-Based Resistive Switching Memories. Nat. Mater. 2007; 6: 833-40. doi: 10.1038/nmat2023
- Shima H, Zhong N, Akinaga H. Switchable Rectifier Built With Pt/TiOx/Pt Trilayer. Appl. Phys. Lett. 2009; 94: 082905.
- Wang C, Jin KJ, Xu ZT, et al. Switchable Diode Effect and Ferroelectric Resistive Switching in Epitaxial BiFeO3 Thin Films. Appl. Phys. Lett. 2011; 98: 192901. doi: 10.1063/1.3589814
- Park C, Chadi D. Microscopic Study of Oxygen-Vacancy Defects in Ferroelectric Perovskites. Phys. Rev. B. 1998; 57: R13961. doi: 10.1103/PhysRevB.57.R13961


