Research Article
Rheological, Thermal, and Foaming Behaviors of Different Polylactide Grades
Department of Metallurgical & Materials Engineering, Faculty of Chemical and Metallurgical Engineering, Istanbul Technical University, Maslak/Istanbul, 34469, Turkey
*Corresponding author: Mohammadreza Nofar, Department of Metallurgical & Materials Engineering, Faculty of Chemical and Metallurgical Engineering, Istanbul Technical University, Maslak/Istanbul, 34469, Turkey, E-mail: nofar@itu.edu.tr
Received: March 30, 2018 Accepted: April 17, 2018 Published: April 23, 2018
Citation: Nofar M. Rheological, Thermal, and Foaming Behaviors of Different Polylactide Grades. Int J Mater Sci Res. 2018; 1(1): 16-22. doi: 10.18689/ijmsr-1000103
Copyright: © 2018 The Author(s). This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
This article compares the rheological, thermal, and foaming behaviors of various polylactide (PLA) grades supplied by NatureWorks and their compounds with three different fillers. The results depicted that linear PLAs with various D-content had similar viscoelastic behavior whereas branching and addition of additives improved PLA's complex viscosity and its elastic behavior. Strain hardening behavior was also observed only in PLA with branched structures. Increased complex viscosity due to the induced crystallization was also expedited in PLAs with lower D-content. Addition of chain extender and additives also expedited the crystallization rate and hence expedited the shear viscosity increase. Among the additives, micro-sized talc enhanced PLA's crystallization rate more effectively than the nano-sized additives due to its more dominant role on crystal growth than crystal nucleation that exist in PLAs with nanoparticles. Foam uniformity was however enhanced more significantly in PLAs with nanoparticles.
Keywords: Polylactide; PLA; Rheology; Crystallinity; D-content; Chain extender; Nanoparticles; Foaming.
Introduction
Polylactide (PLA) is a promising candidate to meet increasing requirements as an environmentally friendly biopolymer in a wide variety of industries. However, PLA's slow crystallization, low melt strength and low heat deflection temperature create a challenge in the mass production of PLA products [1-6]. The melt processing involves heating the polymer beyond its melting temperature, shaping to form the desired product, and cooling with stabilized geometry. Therefore, it is essential to understand the thermal, crystallization, and melt rheological behaviors of the polymer to optimize its processing. The melt rheological properties of a polymer could significantly determine how the melt flows and final product shapes while it could also control the crystallization mechanism [7]. Polymers' rheological properties are highly dependent on temperature, molecular weight and shear rate. According to the melt rheological response of a polymer under various shear rates that correspond to different processing types, the processing temperatures could be defined [8]. The thermal and rheological properties are very determinative parameters in such polymer processes as foaming.
There are only few studies that examined and compared the rheological properties of various PLAs with different D-lactide contents. Although the presence of various D-lactide contents mainly influences the PLA molecular affinities toward crystallization, the melt rheological properties could also illustrate differences as the affinity of molecules in the melt condition could also influence the viscoelastic features of PLA melt. The zero shear viscosity decreases with the D-lactide comonomer content when experiments are conducted between Tg and Tg +100°C [9]. Between this temperature range, cold crystallization could be induced during the measurements and hence for PLAs with low D-lactide comonomer the viscosity could increase as the consequence of cold crystallization. It was also showed that PLAs with some degree of D-content possess lower zero shear viscosity than homo PLLA with no D-lactide [10]. On the other hand, the elongational viscosity of the PLAs with lower D-lactide reveals earlier strain hardening at 180°C. It is suggested that extension thickening appears when the deformation rate considerably exceeds the rate of molecular relaxation. This means that for the branched samples with longer molecular relaxation time the extensional thickening could occur at lower extensional rates [10].
A large number of studies investigated the influence of using various chain extenders in PLA to introduce long chain and branched structures in order to improve the melt strength and viscoelastic behavior of PLA. Dorgan et al. [11] synthesized PLA on-site via generating two-arm (linear) and four and six arms star PLA molecules. They showed that the six-arm star PLA had a higher melt viscosity than the four-arm star for comparable arm molecular weight and the zero shear viscosity increased with branching. Among the chain extenders the successfully investigated case that led to improve the PLA's viscoelastic properties has been Joncryl ADR4368 [12-18]. Joncryl is an epoxy-based multifunctional oligomeric chain extender and has been proved to be one of the effective chain extenders that could not only improve the viscoelastic and rheological properties of PLA but could also improve the PLA's slow crystallization rate and increase its degree of crystallization [19-20]. Wang et al. [14] also illustrated that the addition of Joncryl significantly increased the complex shear and elongational viscosities of PLA with strain hardening behavior and thereby improves the melt strength and crystallization rate of PLA. These improvements could significantly enhance the extrusion foamability of PLA resin and caused great progress in cell density, foam morphology uniformity and foam expansion. The extrusion foamability enhancements via improving the PLA's melt strength (complex shear and elongational viscosities) and its crystallization kinetics through the addition of chain extender was also confirmed by Mihai et al. [21] while they used Cesa Extend OMAN698493 as chain extender. Najafi et al. [16] also revealed that the branching with Joncryl ADR4368 could significantly improve the batch foaming-ability of PLA.
Generally, in foaming, an optimal degree of crystallization is needed to achieve a desirable fine cell structure and a reasonable expansion ratio [6, 22]. Various studies have been done on PLA foaming using various fillers, D-content, PLA blends, branching degree, strain induction, and gas contents. All of these investigations show foaming technology is a wellestablished process while expansion and dissolved blowing agent could improve PLA's crystallization rate and its degree of crystallinity [22-36]. In this study, the rheological and thermal properties of various PLA grades provided from NatureWorks are compared. Subsequently the foaming behavior (the cellular morphology and foam uniformity) of these PLAs with various D-content, linear and branching structure, and nano-/micro-sized additives are compared via solid-state batch foaming.
Experimental Procedure
Materials
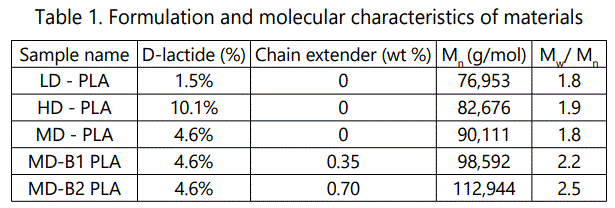
Three commercially available PLA grades, Ingeo™ 3001D, 8051D, 8302D, with D-contents of 1.5, 4.6, and 10.1%, respectively, were supplied by NatureWorks® LLC. In this study, they were referred to as low-D (LD), medium-D (MD), and high-D (HD) linear PLAs. Two branched PLA grades also supplied by NatureWorks® LLC were produced by reactive extrusion with an epoxy-based multi-functional oligomeric chain extender (Joncryl ADR-4368C, BASF Inc.). These branched PLA were based on 8051D (MD-PLA) with 0.35 and 0.7wt% of the chain extender (CE). These samples were also referred to as MD-B1 and MD-B2 PLAs, respectively. The PLA grades' molecular characteristics are shown in Table 1.
Three different additives were also compounded with MD-PLA. These additives were micro-sized talc (Mistron Vapor-R grade) with a mean particle size of 2 μm, nanoclay Cloisite 30B (CN), and nanosilica Aerosil A200 (SiN). These compounds were also referred to as MD-1talc, MD-1SiN, and MD-1CN, respectively.
Sample Preparation
To decrease the moisture level of the PLA pellets and the additives, they were oven-dried at 65°C for at least 6 hours before processing. Using a Minilab twin-screw extruder, the additives were melt-blended at a 1 wt% loading level with PLA material at 180°C for 4 min. with a screw rpm of 40. Neat PLA materials were also processed in the Minilab compounder to maintain a base material with the same history as the PLAadditive compounded samples. Then, the PLA samples were hot-pressed using a hydraulic compression machine (Carver Inc.) at 200°C for 5 minutes to produce circular samples with a diameter of 25 mm and a gap size of 1mm for parallel plate rheological experiments. Extensional viscosity samples were also prepared in a rectangular shape with dimensions of 1cm × 2cm × 1mm.
Rheological tests
Rheological measurements were performed using a TAInstruments ARES rheometer, equipped with a standard force transducer, which can measure a maximum torque of 200 mN.m. Circular hot pressed samples were used to explore PLA's linear viscoelastic behavior in a dynamic frequency sweep test at 180°C and strain of 5%. To investigate the complex viscosity variation through the induced crystallization, dynamic time sweep test in small amplitude oscillatory shearing (SAOS) was also implemented at isothermal melt crystallization temperatures. Also, the extensional viscosity measurements were done at the melting temperature of each PLA sample under strain rates of 0.01, 0.1, and 1.
Polarized optical microscope (POM)
An Olympus BX51 optical microscope equipped with cross polarizer filters and a hot-stage was used to examine the isothermal melt crystallization kinetics (i.e.,crystal nucleation and growth) of PLA with talc and nanoclay at 100°C.
Thermal analysis experiments
A DSC Q2000 (TA Instruments) was used to investigate the crystallization behavior of PLA samples. First, the nonisothermal melting crystallization of PLAs were investigated. Samples were heated from room temperature to 200°C at a heating rate of 20°C/min. and then equilibrated at 200°C for 10 min. to remove all previous thermal and stress histories. Then the samples were cooled to 25°C at a rate of 2°C/min. to allow sufficient diffusion time for the crystallization process. Isothermal melting crystallization experiments of PLAs were also conducted after eliminating their thermal and stress histories as in the non-isothermal experiments. The samples were then cooled at a rate of 20°C/min. to isothermal temperature of 100°C and equilibrated until full crystallization. During all processes, the DSC curves were recorded and analyzed using TA Universal software. The crystallinity of samples was calculated by using the following Equation:

where ΔHc is the crystallization and 93.6 is the melting enthalpies in J/g of 100% crystalline PLA [37].
Solid-State Batch Foaming
Batch foaming experiments were carried out in a highpressure vessel and saturated with CO2 at a pressure of 1500 psi at the ambient temperature 25°C for 24 hours. Then, the saturated samples were taken out of the chamber immediately following a rapid release of the pressure. They were then dipped in a hot silicon oil bath at 150°C for 60 seconds before being quenched in cold water to stabilize their foam morphology. The samples' foaming was triggered by the rapid pressure drop and temperature rise.
Results and Discussion
Rheological behaviors
PLA's melt rheological properties could significantly affect how the polymer flows during the different processing techniques. The PLA rheological properties are highly dependent on temperature, molecular weight and shear rate. Moreover, by branching the PLA's molecules not only the molecular weight would increase but also the molecular entanglement generated by branching could profoundly influence the melt rheological properties of PLA. The melts of high molecular weight PLA behave like a pseudoplastic, nonNewtonian fluid. In contrast, low molecular weight PLA shows Newtonian-like behavior at shear rates. Also, as shear rates increase, the melt viscosities decrease noticeably and hence the polymer melt exhibits shear-thinning behavior. Moreover, semi-crystalline PLA samples tend to possess higher viscosity than amorphous samples and, hence, when the D-lactide content decreases, higher melt viscosity could be expected as the crystallization capability increases [7].
The rheological behaviors (i.e., the complex and elongational viscosities) of the PLA samples with different branching degrees, D-lactide contents, and presence of 1 wt% nano-/micro-sized additives are shown in Figure 1. As seen, the complex viscosity of PLA at 180°C significantly increased with an increase in the branching degree and with addition of 1 wt% additives. This increase was almost 1 to 2 orders of magnitude. MD-linear PLA and its composites revealed a Newtonian behavior along a large range of low frequencies, and at high frequencies a shear-thinning behavior was observed. In branched samples the shearthinning behavior is more dominant, and at low-frequencies the complex viscosity further increases due to the shape relaxation and network formation of the branched molecular structure.
In the case of PLAs with various D-contents, the HD-PLA possesses a higher viscosity which is most probably due to its higher molecular weight as reported in Table 1. On the other hand, the LD-PLA with a lower molecular weight depicts a higher viscosity than the MD-PLA which could be due to the fact that PLA with high crystallization capability could possess higher viscosity specifically at processing temperatures right beyond their melting temperature.
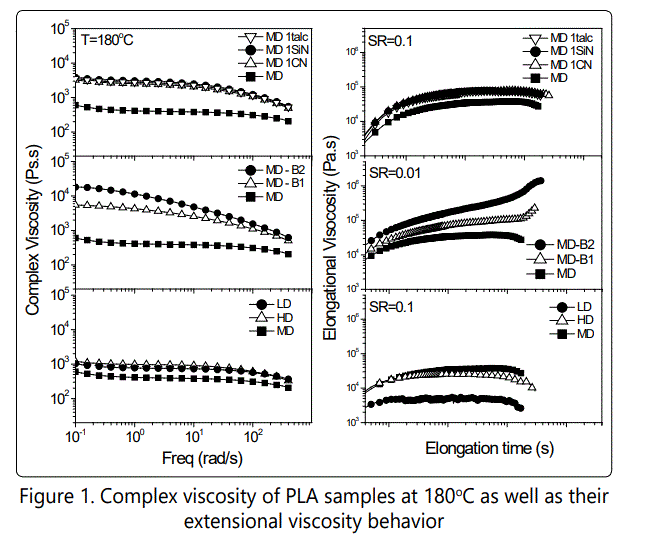
On the other hand, the elongational viscosity of the PLA samples shows a strain hardening behavior only in the branched PLA. Table 2 also shows the rheological characteristics of different PLA materials such as the relaxation time and the zero shear viscosity.
The isothermal melt crystallization behavior of the PLA samples was also investigated using the SAOS experiments and in this case the increase in complex viscosity reflects the induction of crystallization and hence the amount of increase could resemble the amount of induced crystallinity qualitatively. As mentioned, in order to properly compare all of the PLA samples, all grades were passed through the processing that the PLA compounds were passed through. Therefore, for neat grades an extra processing has been conducted. In order to see if the applied processing could have modified the properties of the PLA samples, firstly the isothermal melt crystallization of MD-PLA was analyzed using SAOS experiment. Figure 2 shows that processed MD-PLA reveals lower initial viscosity, however, the isothermal crystal induction expedites compared to that in "as received" MDPLA (MD AR). This could be due to the possible molecular degradation of PLA sample during the processing which first of all reduces the initial complex viscosity and subsequently causes faster crystallization due to existence of shorter molecules and hence easier crystallizability. The amount of increase in complex viscosity also illustrates that larger content of isothermal crystallinity was formed in the processed MD-PLA.

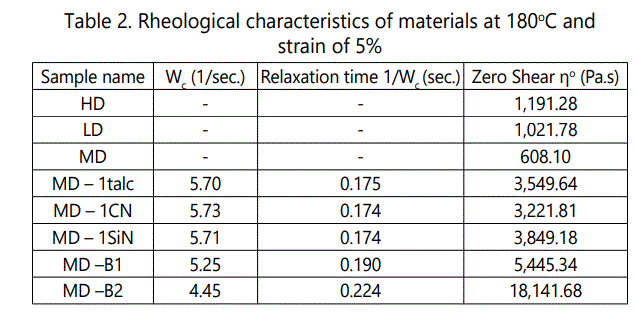
Figure 3a also illustrates how the isothermal melt crystallization of the processed MD-PLA varies with isothermal temperature. The initial viscosity of the samples decreases with increase of the isothermal temperature due to the reduced melt strength and increased molecular mobility. On the other hand, 100°C is the temperature at which the crystallization rate is the fastest and above and below this temperature, the crystal growth and crystal nucleation are the dominated crystallization mechanisms, respectively [31-32]. According the normalized results shown in Figure 3b, the amount of increase in complex viscosity also illustrates that larger content of isothermal crystallinity was formed at the order of 100, 110, 90 and then 120°C.
Figure 4 shows the isothermal melt crystallization of all PLA grades explained in this study through SAOS experiments at 100°C. The initial viscosity results of the samples are consistent with the results showed in Figure 1 for PLAs with different D-lactide content, with different branching degrees and with various fillers. The increase in viscosity due to the induction of crystallinity however, shows that in PLAs with various D-contents, the LD-PLA crystallizes very quickly and HD-PLA not only does not crystallize but also started to degrade as is being kept during the isothermal condition due to thermal degradation.
On the other hand, in branched PLAs, with the increase in branching degree the crystallization rate increases as the crystallization occurs at earlier times however, the amount of crystallinity (reflected by the amount of viscosity increase during isothermal annealing) decreases. The earlier is because the branched molecules actively behave as crystal nucleating sites and hence the crystallization could occur much faster [20] and the later is due to the hindered molecular mobility in branched cases that limits the crystal growth and thereby the final crystallinity. The hindered mobility is not only because of the branching but also due to the increase in crystal nucleation in the branched cases. This fastened crystal nucleation rate already increases the molecular entanglement and limits the crystal growth mechanism.
In the PLA samples with various fillers, as Figure 1 illustrated, the initial viscosity of the PLA samples with various fillers doesn't change significantly and the amount of increase in complex viscosity seems very similar for all three PLA compounds reflecting that similar amount of crystallinity has been induced in all three systems.
However, the micro-sized talc acts more actively in promoting the crystallization rate of PLA compared to those of nanosilica and nanoclay. The crystallization mechanism is more dominated with crystal growth in PLA with talc whereas in PLA-nanoclay and PLA-nanosilica cases the crystallization is more dominated with crystal nucleation. This could also be proven with the polarized optical microscopy results done for PLA-talc and PLA-nanoclay at 100°C isothermal condition in Figure 5.

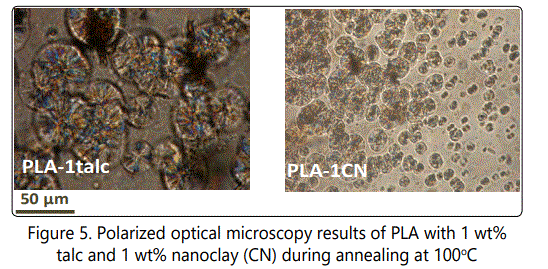
Thermal behaviours
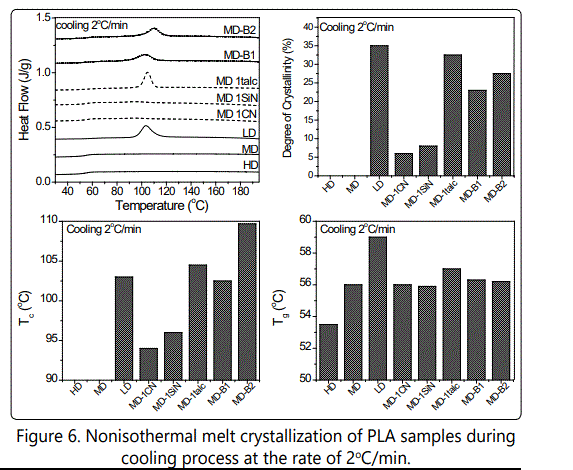
Non isothermal melt crystallization of various PLA grades was examined using DSC experiments during cooling at a rate of 2o C/min. Figure 6 compares the cooling graphs, obtained degree of crystallinity, crystallization and glass transition temperatures. Among the PLAs with various D-contents, only LD-PLA could crystallize during such a slow cooling rate. On the other hand, as noted in section 1 of the results and in our previous study [20] the branching expedited the crystallization of the samples and as the chain extender content increases the crystallization occurs earlier and the amount of crystallinity also increases. On the other hand, similar to the results of section 1, talc fastened the crystallization of MD-PLA and compared to nano-additives, much larger degree of crystallinity could induce with talc (~32 %) whereas this value for MD-PLA with nanoparticles was below 10 %. The crystallization also occurred much earlier in PLA compound with talc than those with nanosilica and nanoclay. In all, the LD-PLA, MD-B2, and MD-1talc revealed more expedited crystallization behavior compared to other PLA samples and compounds. This was also confirmed by isothermal melt crystallization of PLA samples that were conducted in DSC at 100o C. As seen these results are also consistent with the SAOS isothermal annealing results that were illustrated in section 1. The glass transition temperatures (Tg) of the PLA grades show that the Tg values increases with decrease in D-content due to the existence of crystallites that further limit the regularity of the amorphous region. The branching and the addition of fillers however didn't reveal a significant influence on PLA's Tg.

Solid-State Batch Foaming behaviors
The batch foaming behaviour of various PLA samples is depicted in Figure 8. The LD-PLA couldn't show a uniform foaming behavior due to the very fast crystallization that prevents the cell growth and expansion. So no cellular morphology could be observed. On the other hand, low melt strength could have quickly caused the coalescence of any possible generated bubbles. As the D-content increases, the stiffness caused by crystallization was reduced and hence the nucleated bubbles could grow and expand easier however, lack of sufficient melt strength did not let to have a uniform foam morphology in both MD and HD PLAs and hence the chance of cell coalescence is still high.
On the other hand, the induction of branching could improve the melt strength during the foaming and hence with addition of chain extender, the cell morphology looks more uniform most probably due to the reduced degree of cell coalescence.
The PLA compounds however, revealed a more uniform foam morphology and that was more prominent in the compounds with nanoparticles. In the case of MD-PLA with talc, the non-uniform foam morphology could be due to the generation of large size crystals that could expel some dissolved gas and hence around these crystals more bubble nucleation could appear and causes this bimodal non-uniform morphology [38]. On the other hand, the PLA samples with nanoparticles revealed much more uniform foam morphology within the range of microcellular foams which possess average cell sizes below 30 μm [39-40]. The increase cell density in presence of well dispersed nanoparticles is firstly due to the heterogeneous cell nucleation around these nanoparticles and then is most likely due to the similar heterogeneous cell nucleation but also around the large number of nucleated crystals that the nanoparticles promoted their generation. Both PLA compounds with nanosilica and nanoclay revealed similar cell morphology and this means that through the current solid-state batch foaming not much differences could be appeared for these compounds with nanoparticles possessing various geometries.

Conclusion
The rheological, thermal and foaming behavior of PLA samples based on PLA molecules with different D-lactide contents, with linear and branched structures, and in presence of micro-/nano-sized additives were investigated in this study. The viscoelastic properties of these PLA were explored within their linear viscoelastic region through small amplitude oscillation shearing (SAOS). The complex shear viscosity variations caused by induced crystallization was also investigated during isothermal conditions. The extensional viscosity of these grades were also compared. The non-isothermal and isothermal melt crystallization behaviors of the noted PLA grades were investigated using a differential scanning calorimeter (DSC). The foaming behavior of these PLA samples were also compared using solidstate batch foaming in a pressure vessel.
The results revealed that additives and branching could improve the complex shear viscosity of PLA samples, whereas branching could further improve the strain-hardening behavior of PLA. The crystallization rate and the degree of crystallinity of the LD-PLA, MD-PLA with talc and chain extender were the highest among all samples. However, MDPLA samples with nanoparticles showed more improvement of crystal nucleation mechanism during the crystallization. The larger number of nucleated crystals together with the well dispersed nanoparticles improved the foam uniformity and cell nucleation behavior of PLA samples dramatically.
Acknowledgements
The author would like to acknowledge the financial supports of Istanbul Technical University Scientific Research Projects (ITUBAP) with the project numbers of 39415 and 40616.References
- Garlotta DJ. A literature review of poly(lactic acid). J Polym Environ. 2001; 9(2): 63-84. doi: 10.1023/A:1020200822435
- Sinclair RG. The case for polylactic acid as a commodity packaging plastic. J Macromol Sci Part A Pure Appl Chemi. 1996; 33(5): 585-597. doi: 10.1080/10601329608010880
- Grijpma DW. Pennings AJ. (Co)polymers of L-lactide, 2. Mechanical properties. Macromol Chem Phys. 1994; 195(5): 1649-1663. doi: 10.1002/ macp.1994.021950516
- Auras R, Harte B, Selke S. An overview of polylactides as packaging materials. Macromol Biosci. 2004; 4(9): 835-864. doi: 10.1002/mabi.200400043
- Drumright RE, Gruber PR, Henton DE. Polylactic acid technology. Adv Mater. 2000; 12(23): 1841-1846. doi: 10.1002/1521-4095(200012)12;23<1841::AIDADMA1842>3.0.CO;2-E.
- Nofar M, Park CB. Poly (lactic acid) foaming. Prog Polym Sci. 2014; 39(10): 1721-1742. doi: 10.1016/j.progpolymsci.2014.04.001
- Lim LT, Auras R, Rubino M. Processing technologies for poly (lactic acid). Prog Polym Sci. 2008; 33(8): 820. doi: 10.1016/j.progpolymsci.2008.05.004
- Osswald, Tim A, Rudolph, Natalie. Polymer rheology: fundamentals and applications. Publisher: Hanser Publications. 2015; ISBN: 978-1-56990- 517-3. doi: 10.3139/9781569905234
- Palade L-I, Lehermeier h J, Dorgan JR. Melt rheology of high l-content poly(lactic acid). Macromolecules. 2001; 34(5): 1384-1390. doi: 10.1021/ma001173b
- Dorgan J, Williams J. Melt rheology of poly(lactic acid), Entanglement and chain architecture effects. J Rheol. 1999; 43(5): 1141. doi: 10.1122/1.551041
- Meng, Q. Heuzey, M.C. Carreau, P. Wood-Adams, P.M. Control of thermal degradation of polylactide/clay nanocomposites during melt processing by chain extension reaction. Polym. Degrad Stabil. 2012; 97(10): 2010- 2020. doi: 10.1016/j.polymdegradstab.2012.01.030
- Najafi N, Heuzey M.C, Carreau P, Wood-Adams, P.M. Control of thermal degradation of polylactide (PLA)-clay nanocomposites using chain extenders. Polym. Degrad. Stabil. 2012; 97 (4): 554-565. doi: 10.1016/j. polymdegradstab.2012.01.016
- Wang J, Zhu W, Zhang H, Park CB. Continuous processing of low-density, microcellular poly(lactic acid) foams with controlled cell morphology and crystallinity. Chem Eng Sci. 2012; 75: 390-399. doi: 10.1016/j.ces.2012.02.051
- Al-Itry R, Lamnawar K, Maazouz, A. Reactive extrusion of PLA, PBAT with a multi-functional epoxide: Physico-chemical and rheological properties. Eur Polym J. 2014; 58: 90-102. doi: 10.1016/j.eurpolymj.2014.06.013
- Najafi N, Heauzey MC, Carreau PJ, Therriault D, Park CB, et al. Rheological and foaming behavior of linear and branched polylactides. Rheol Acta. 2014; 53(10): 779-790. doi: 10.1007/s00397-014-0801-3
- Gu, L. Xu, Y. Fahnhorst, G. W. Macosko, C. W. Star vs long chain branching of poly(lactic acid) with multifunctional aziridine. Journal of Rheology. 2018; 61(4): 785-796. doi: 10.1122/1.4985344
- Nofar M, Zhu W, Park CB. Effect of dissolved CO2 on the crystallization behavior of linear and branched PLA. Polymer. 2012; 53 (15): 3341-3353. doi: 10.1016/j.polymer.2012.04.054
- Nofar M, Zhu W, Park CB, Randall J. Crystallization kinetics of linear and long-chain-branched polylactide. Ind Eng Chem Res. 2011; 50(24): 13789- 13798. doi: 10.1021/ie2011966
- Mihai M, Huneault MA, Favis BD. Rheology and extrusion foaming of chain-branched poly(lactic acid). Polym Eng Sc.i. 2010; 50(3): 629-642. doi: 10.1002/pen.21561
- Nofar M. Effects of nano-/micro-sized additives and the corresponding induced crystallinity on the extrusion foaming behavior of PLA using supercritical CO2 . Mater Design. 2016; 101: 24-34. doi: 10.1016/j. matdes.2016.03.147
- Kesht kar M, Nofar M, Park CB, Carreau PJ. Extruded PLA/Clay Nanocomposite Foams Blown with Supercritical CO2 . Polymer. 2014; 55: 4077. doi: 10.1016/j.polymer.2014.06.059
- Ameli A, Jahani D, Nofar M, Jung PU, Park CB et al. Processing and Characterization of Solid and Foamed Injection-Molded Polylactide with Talc. 2013; 49(4): 351-374. doi: 10.1177/0021955X13481993
- Ameli A, Nofar M, Jahani D, Park CB. Development of high void fraction polylactide composite foams using injection molding: Mechanical and thermal insulation properties. Compos Sci Technol. 2014; 90: 88-95. doi: 10.1016/j.compscitech.2013.10.019
- Ameli A, Nofar M, Jahani D, Park CB. Development of High Void Fraction Polylactide Composite Foams Using Injection Molding: Crystallization and Foaming Behaviors. Chem Eng J. 2015; 262: 78-87. doi: 10.1016/j. cej.2014.09.087
- Nofar M, Ameli A, Park CB. Development of Polylactide Bead Foam with Double Crystal Melting Peak Structure. Polymer. 2015; 69(1): 83-94. doi: 10.1016/j.polymer.2015.05.048
- Nofar M, Ameli A, Park CB. A Novel Technology to Manufacture Biodegradable Polylactide Bead Foam Products. Mater Design. 2015; 83: 413-421. doi: 10.1016/j.matdes.2015.06.052
- Nofar M, Tabatabaei A, Ameli A, Park CB. Comparison of melting and crystallization behaviors of polylactide under high-pressure CO2, N2, and He. Polymer. 2013; 54(23): 6471-6478. doi: 10.1016/j.polymer.2013.09.044
- Nofar M, Ameli A, Park CB. The thermal behavior of polylactide with different D-lactide content in the presence of dissolved CO2 . Macromol Mater Eng. 2014; 299(10): 1232-1239. doi: 10.1002/mame.201300474
- Nofar M, Tabatabaei A, Park CB. Effects of nano-/micro-sized additives on the crystallization behaviors of PLA and PLA/CO2 mixtures. Polymer. 2013; 54(9): 2382-2391. doi: 10.1016/j.polymer.2013.02.049
- Liao X, Nawaby AV. Solvent Free Generation of Open and Skinless Foam in Poly(L-lactic acid)/Poly(D,L-lactic acid) Blends Using Carbon Dioxide. Ind Eng Chem Res. 2012; 51(19): 6722-6730. doi: 10.1021/ie3000997
- Nofar M, Tabatabaei A, Sojoudi H, Park CB, Carreau PJ, et al. Mechanical and bead foaming behavior of PLA-PBAT and PLA-PBSA blends with different morphologies. Eur Polym J. 2017; 90: 231-244. doi: 10.1016/j. eurpolymj.2017.03.031
- Fischer E, Sterzel H, Wegner, H.J. G., Collo. K, et al. Investigation of the Structure Of Solution Grown Crystals of Lactide Copolymers by Means of Chemical Reaction. Polym. Sci. 1973; 251(11): 980-990. doi: 10.1007/ BF01498927
- Taki K, Kitano D, Ohshima M. Effect of growing crystalline phase on bubble nucleation in poly(L-Lactide)/CO2 batch foaming. Ind Eng Chem Res. 2011; 50(6): 3247-3252. doi: 10.1021/ie101637f
- Colton JS, Suh NP. The nucleation of microcellular thermoplastic foam with additives: Part I: Theoretical considerations. Polym Eng Sci. 1987; 27(7): 485-492. doi : 10.1002/pen.760270702
- Colton JS, Suh NP. The nucleation of microcellular thermoplastic foam with additives: Part II: Experimental results and discussion. Polym Eng Sci. 1987; 27(7): 493-499. doi: 10.1002/pen.760270703



