Case Report
Clear Cell Hidradenoma of the Inguinal Area: A Rare Tumor in an Uncommon Location
1Department of Dermatology, University hospital Hassan II, Fez, Morocco
2Hassan Center of Anatomopathology, Rabat, Morocco
*Corresponding author: Safae Zinoune, Doctor, Department of Dermatology, Hassan II University Hospital, Fès, Morocco, E-mail: dr.zinounesafae@gmail.com
Received: April 3, 2019 Accepted: April 8, 2019 Published: April 18, 2019
Citation: Zinoune S, Douhi Z, Baybay H, Elloudi S, Rimani M, Mernissi FZ. Clear Cell Hidradenoma of the Inguinal Area: A Rare Tumor in an Uncommon Location. Madridge J Case Rep Stud. 2019; 3(2): 140-142. doi: 10.18689/mjcrs-1000136
Copyright: © 2019 The Author(s). This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Nodular hidradenomas are benign sweat gland-derived tumors that can produce clinical and dermatoscopic misdiagnoses. Histologically, these tumors can be classified into eccrine and apocrine subtypes. Clear cell hidradenoma is a rare benign eccrine tumor, itʼs representing 1% of all skin tumors, found mainly in adult females. The lesions are usually solitary and are most likely to be found on the scalp, face or anterior trunk. We report a case of clear cell hidradenoma developing in the inguinal area in a 68-year old man, which occurred during the course of nine months. The clinical and histologic findings of the tumor are documented. The lesion was totally removed by excision and revealed no evidence of recurrence. Through this case, we would like to highlight the unusual location of this hidradenoma.
Keywords: Adnexial tumor; Hidradenoma; Eccrine; Clear cells; Groin.
Introduction
Clear cell hidradenoma (CCH) is an uncommon benign tumor of eccrine glands and a variant of hidradenoma first described by Liu in 1949 [1]. This tumor found mainly in adults and is excised more commonly in women than in men. Lesions in children are very rare [2,3]. They are usually seen as solitary, well-defined, non-encapsulated, slowly growing tumor in the dermal layer of head, face, and extremities; rarely show aggressive clinical behavior and malignant transformation [4]. Very few cases of groin CCH are documented in the literature. We present a rare case of CCH in a 65-year-old man in the left inguinal area.
Case Report
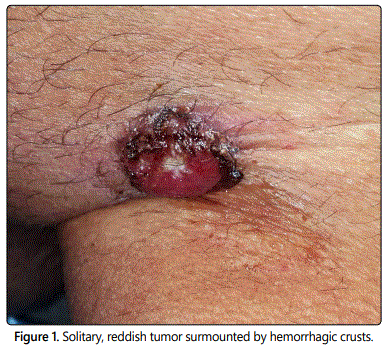
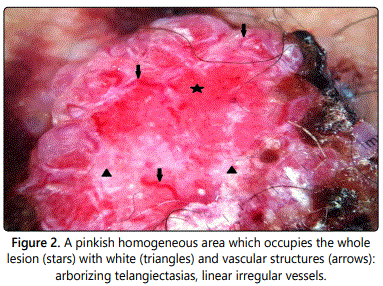
A 68-year-old man presented with a solitary, asymptomatic tumor on the left inguinal area. About one year ago, the patient noticed a papule in this area, but due to being small and without symptoms, no action for diagnosis and treatment was taken. About six months ago, the lesion has started to grow quickly and gradually had become a tumor (Figure 1). Clinical examination revealed a solitary, reddish, firm, bleeding and well-defined, tumor, measuring around 4 × 2.5 cm and surmounted by hemorrhagic crusts. This led to the clinical diagnosis of a squamous cell carcinoma. Other differential diagnoses included acromic melanoma and basal cell carcinoma. Dermoscopy of the tumor showed a pinkish homogeneous area which occupies the whole lesion with white and vascular structures (arborizing telangiectasias, dotted and linear irregular vessels) (Figure 2). Clinically, he had no palpable groin lymphadenopathy and no other significant abnormalities at the somatic examination.
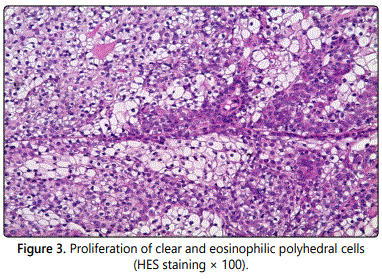
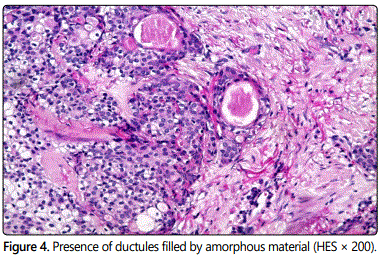
The mass was excised with the overlying skin under local anesthesia. Histopathologic examination with hematoxylineosin staining revealed a multilobulated tumor with solid and cystic components. The tumor showed a composition of clear and eosinophilic cells with ductal differentiation (Figures 3 and 4). No recurrence has occurred at 8 months of follow-up.
Discussion
CCH is a benign tumor of skin appendage that arises from the distal excretory duct of eccrine sweat glands on the skin of head, face, and upper extremities [4]. It is first termed as eccrine acrospiroma, where “spiroma” means adenoma of sweat glands and “acro” means the topmost or end [1]. Other terminologies such as CCH, nodular hidradenoma or solid and cystic hidradenoma are also preferred [5]. CCH occurs at any age but usually seen in 4th-8th decade of life and are twice more common in females [6]. It presents as a single, small, slow-growing, well-demarcated, intra-dermal tumor that varies in size from 5 to 30 mm. The overlying skin varies in color, with a smooth, thickened, atrophic, or ulcerated surface [7]. It is common on the head, neck, and anterior trunk, and infrequent in the lower extremities [1,7]. In our case, a male patient of old age presented with inguinal asymptomatic bleeding tumor. Inguinal region is not a common site for this condition and very few cases have been reported [8,9].
CCH may be asymptomatic or may present with pain, or bleeding, and rarely can show malignant transformation [10]. Ulcerated lesions may resemble squamous cell carcinoma, basal cell carcinoma metastatic disease or other adnexal tumors [11-13]. Dermoscopy shows a nodule of amorphous, white-pink areas, red lacuna-like areas, eccentric ulceration, as well as hairpin, dotted, and linear irregular vessels [14].
Diagnosis is made by histopathology. Histopathological appearance of CCH is very typical, characterized by two types of cells-polyhedral eosinophilic cells and clear cells along with duct-like cystic spaces lined by low cuboidal cells [15]. Focal squamous differentiation may be seen [16], which was not observed in the present case. Malignant transformation of CCH is rare and is marked clinically by a rapid growth with cutaneous ulceration; and histologically by nuclear atypia, increased number of mitotic figures, infiltrative pattern, angiolymphatic or perineural invasion with areas of necrosis [6,17]. Routine hematoxylin and eosin stained sections are usually sufficient for diagnosis and immunohistochemical analysis is not required. Immunohistochemical analysis may become essential in distinguishing CCH from its mimics that may include both benign and malignant lesions. Biernat et al. showed a cytokeratin (CK) expression in CCH. Most keratin expression such as CK6/18, CK7, and CK8/18 was noted in squamoid cells and tubule lining cells. Clear cells were consistently positive for epithelial membrane antigen and CK10/17/18 [13]. Search in literature showed a single case of nodular hidradenoma in inguinal lymph node reported [8]. But in our case, the inguinal lymph node was not involved.
The usual treatment of choice of the tumor is wide surgical excision to minimize the risk of local recurrence [18]. In our patient, the tumor was very atypical in terms of its size and ulcerated surface, thus making malignancy a possibility especially considering the advanced age of the patient. Excision of the mass was a difficult task as the nodule was very vascular and its seat at the inguinal area. It was important to exclude this as there is a small risk in such tumors of a malignant transformation into hidradenocarcinoma. Surprisingly, the histology in this patient showed bland cytology despite the size and rich vasculature of the tumor.
Conclusion
Inguinal CCH is a rare benign tumor that remains a challenging entity to diagnose clinically without the presence of any distinctive clinical or dermoscopic features. Though these tumors are usually benign, there is always a concern that they could be malignant. It should be considered as a differential diagnosis when encountering ulcerated tumors.
References
- Liu Y. The histogenesis of clear cell papillary carcinoma of the skin. Am J Pathol. 1949; 25: 93-103.
- Winkelmann RK, Wolff K. Solid–cystic hidradenoma of the skin. Clinical and histopathologic study. Arch Dermatol. 1968; 97(6): 651-661.
- Faulhaber D, Worle B, Trautner B, Sander C. Clear cell hidradenoma in a young girl. J Am Acad Dermatol. 2000; 42(4): 693-695. doi: 10.1016/S0190-9622(00)90192-0
- Paranjyothi M, Mukunda A. Clear cell hidradenoma: An unusual tumor of the oral cavity. J Oral Maxillofac Pathol. 2013; 17(1): 136-138. doi: 10.4103/0973-029X.110719
- Kaur S, Amanjeet, Thami GP, Mohan H. Intralesional steroid induced histological changes in the skin. Indian J Dermatol Venereol Leprol. 2003; 69: 232-234.
- Stratigos AJ, Olbricht S, Kwan TH, Bowers KE. Nodular hidradenoma: report of three cases and review of the literature. Dermatol Surg. 1998; 24(3): 387-391.
- Tingaud C, Costes V, Frouin E, et al. Lymph node location of a clear cell hidradenoma: report of a patient and review of literature. J Cutan Pathol. 2016; 43(8): 702-706. doi: 10.1111/cup.12720
- Rekhi B, Ingle A, Patil B, Jambhekar NA. Cytomorphological spectrum of a nodular hidradenoma in a young girl presenting with an inguinal soft tissue mass. Cytopathology 2011; 22(2): 135-137. doi: 10.1111/j.1365-2303.2010.00792.x
- Diab M, Gabali A, Kittaneh M. Malignant acrospiroma: a case report in the era of next generation sequencing. BMC Cancer. 2017; 17(1): 221. doi: 10.1186/s12885-017-3217-5
- Nakazato S, Yanagi T, Inamura Y, Kitamura S, Hata H, Shimizu H. A case of nodular hidradenoma of the auricle mimicking squamous cell carcinoma. Eur J Dermatol. 2016; 26(4): 409-410. doi: 10.1684/ejd.2016.2810
- Shahmoradi Z, Mokhtari F. Clear cell hidradenoma. Adv Biomed Res. 2013; 2: 40. doi: 10.4103/2277-9175.109742
- Volmar KE, Cummings TJ, Wang WH, Creager AJ, Tyler DS, Xie HB. Clear Cell Hidradenoma A Mimic of Metastatic Clear Cell Tumors. Arch Pathol Lab Med. 2005; 129(5): e113-e116. doi: 10.1043/1543-2165(2005)129<e113:CCHAMO>2.0.CO;2
- Robles-Mendez JC, Martinez-Cabriales SA, Villarreal-Martínez A, et al. Nodular hidradenoma: dermoscopic presentation. J Am Acad Dermatol. 2017; 76(2): S46-S48. doi: 10.1016/j.jaad.2016.07.003
- Hernández-Pérez E, Cestoni-Parducci R. Nodular hidradenoma and hidradeno carcinoma. J Am Acad Dermatol. 1985; 12: 15-20. doi: 10.1016/S0190-9622(85)70002-3
- Stanley RJ, Sanchez NP, Massa MC, Cooper AJ, Crotty CP, Winkelmann RK. Epidermoid hidradenoma. A clinicopathologic study. J Cutan Pathol. 1982; 9(5): 293-302.
- Biddlestone LR, Mclaren KM, Tidman MJ. Malignant hidradenoma: A case report demonstrating insidious histological and clinical progression. Clin Exp Dermatol. 1991; 16: 474-477.
- Sirinoglu H, Çelebiler Ö. Benign nodular hidradenoma of the face. J Craniofac Surg. 2011; 22(2): 750-751. doi: 10.1097/SCS.0b013e3182085501




