Case Report
Granulomatous Mycosis Fungoides Presented as Multiple Subcutaneous Masses: a case of 5-year follow-up and Review of the Literature
1Department of Dermatology, Tongren Hospital Shanghai Jiao Tong University School of Medicine, Shanghai, P.R. China
2Department of Dermatology, Jiangsu Province Geriatric Hospital, Nanjing, P.R. China
3Department of Dermatopathology, Shanghai Skin Diseases Hospital, Shanghai, P.R. China
4Department of General surgery, The Shibei hospital of Jingan district, Shanghai, P.R. China
*Corresponding author: Song Ning-Jing, Tongren Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, P.R.China, E-mail: songnj-10@163.com
Cheng Xing-Wang, The Shibei hospital, Jingan district, Shanghai, P.R.China, E-mail: chengxingwangbb@163.com
Received: December 25, 2017 Accepted: January 29, 2018 Published: February 05, 2018
Citation: Fang Y, Xiao-Yun X, Jia C, Ning-Jing S, Xing-Wang C. Granulomatous Mycosis Fungoides Presented as Multiple Subcutaneous Masses: a case of 5-year follow-up and Review of the Literature. Madridge J Cancer Stud Res. 2018; 2(1): 34-39. doi: 10.18689/mjcsr-1000104
Copyright: © 2018 The Author(s). This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Summary
Diagnosis of granulomatous mycosis fungoides is very challenging due to its clinical and pathological heterogenicity. We report a case of a 72-year-old female presented with 2 years scaly plaques, 5 months subcutaneous tumours on the sternum and left upper and both side lower extremities.
Keywords: Granulomatous Mycosis Fungoides; Histology; Treatment
Introduction
Granulomatous mycosis fungoides (GMF) is a rare histological variant of cutaneous T-cell lymphoma (CTCL) [1], which was first described by Ackerman and Flaxman in 1970 [1]. GMF usually presented with non-specific skin lesions such as papule, macule, plaque, desquamation asymptomatic nodule, ulcer or tumor. However, the case of ichthyosiform eruption and subcutaneous mass has been reported rarely. We summarize a case of GMF a 72-year-old female initially presented with scaly plaques and subcutaneous tumors, which progressed to a tumoral cutaneous stage with sternum, anterior abdominal and axillary lymph nodes involvement.
Report
A 72-year-old woman was referred to our clinic center in August 2012 presented with 2 years scaly plaques, 5 months subcutaneous masses on the sternum and left upper and both side lower extremities. The patient noticed scaly plaques with mild itching on the right lower limb in April 2010. She was hospitalized twice due to ichthyosis in April and June 2010. Ultrasound revealed enlarged lymph nodes in the subclavian, armpit, and groin. Infiltrative plaques appeared on the surface of her right thigh with no obvious symptoms initially, and sooner, similar symptoms and lumps successively emerged on the sternum and left upper and both side lower extremities but no burst. Six months later she was re-admitted. Biopsies showed granuloma formation in the dermis and subcutaneous tissue, with less heteromorphic cells and more giant cells. Immunohistochemical staining revealed that the tumor cells expressed CD3, CD45RO, but they were negative in L26, CD79a, Bcl-2. Some of atypical cells were CD4⁺ and CD8⁺. T-cell receptor (TCR) gene analysis showed negative expression. Combiningmorphology and immunohistochemical staining with cloned gene rearrangement detection results, there was no certain malignant evidence. However, the rash still had no clear improvement and the tumor continued to grow, so she came to our clinic to seek further medical treatment. Further questions about the disease revealed a subcutaneous mass history in her left pinkie without symptoms in September 2008and then spread to her left ring finger. Histopathological recorded as chronic granuloma formation in local hospital and her two fingers were amputated in October 2009.
Clinical Findings
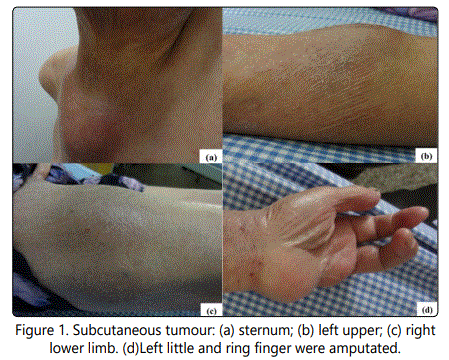
Infiltrative dark erythematous plaques and desquamation presented in most surface skin in cutaneous examination. Subcutaneous tumors were found on the sternum, left upper and both side lower extremities (Fig.1 a,b,c) measuring from 2-3 cm to 6-7cm. Left little and ring finger were amputated (Fig.1d).
Ultrasound Examination
Subcutaneous masses appear in both ears, thighs and right anterior chest wall. Lymph node enlargement, especially in the neck and both groin.
Computed Tomography Scan
The CT scans of the thorax, abdomen and pelvis demonstrated extensive involvements of sternum, anterior abdominal and axillary lymphadenopathy.
Histology and Immunohistology
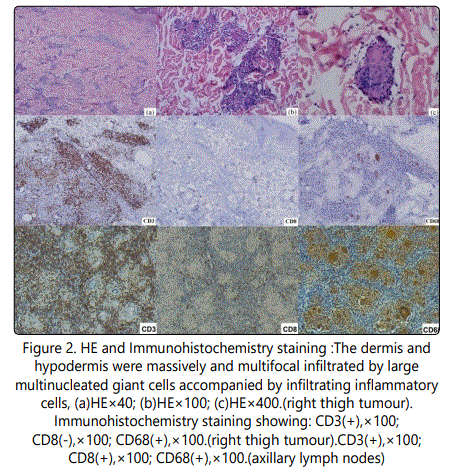
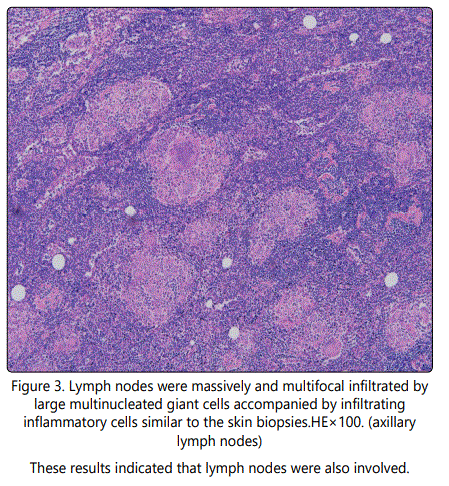
Histologically, the right thigh tumor showed that the dermis and hypodermis were massively and multifocal infiltrated by large multinucleated giant cells accompanied by inflammatory cells such as lymphocytes, histocytes and plasma cells (Fig.2). The neoplastic cells displayed a immunophenotype of CD3⁺,CD4⁺,CD5⁺, CD45RO⁺,CD68⁺,S-100⁺,Ki-67- ,PCNA⁻,LCA⁻, CD8⁻, CD79a⁻, CD20⁻, CD30⁻, CD56⁻, CD1a- (Fig.2) pattern. Immunohistology of axillary lymph node revealed similar results to the right thigh tumor (Fig.2, Fig.3).
TCR Gene Analysis
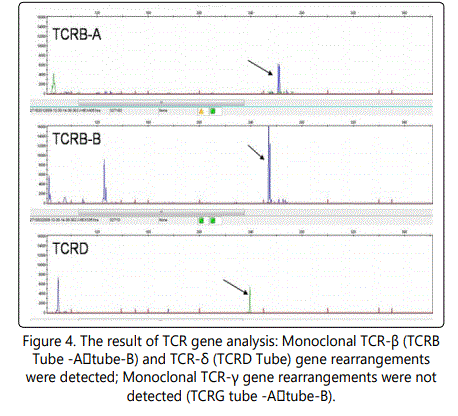
TCR gene analysis of the right thigh biopsy specimen displayed TCRB⁺, TCRD⁺, TCRG⁻ (Fig.4), providing evidence of a T-cell monoclonal proliferation.
Diagnosis
With the combination of clinical presentation, histopathology, immune histology and TCR gene analysis, the diagnosis of granulomatous mycosis Fungoides (GMF) tumor-stage with lymph node involvement was made.
Treatment and Prognosis
The patient was treated with prednisone 20mg and isotretinoin 20mg per day and moisturizer for external use. The masses were gradually disappeared one month after the treatments. The medication doses were tapered off due to the disease progression. Follow-up has been continuing.
Discussing
Granulomatous mycosis fungoides was first described by Ackerman and Flaxman in 1970, who reported a patient with tumor-stage MF with giant cells dermal infiltration [1]. It is a rare histopathological type of mycosis Fungoides (MF) which exhibited characteristic expression of granulomatous reaction with the atypical lymphocytic infiltration [1]. The clinical manifestations of GMF were lack of specificity, therefore GMF cannot be simply diagnosed from the skin rash. It also needs additional tests, such as histopathology, immune histology and TCR gene analysis.
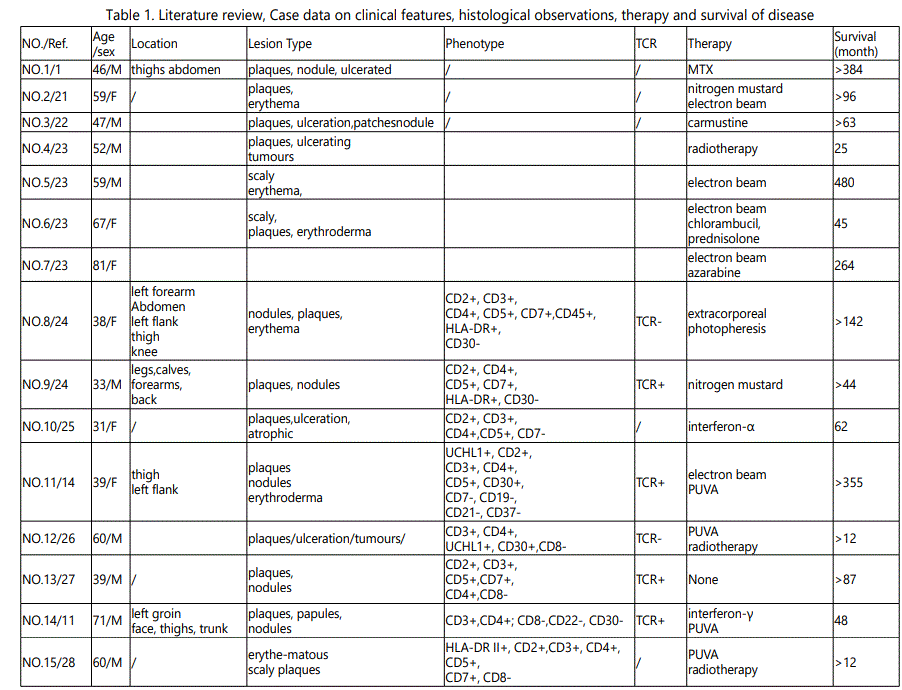
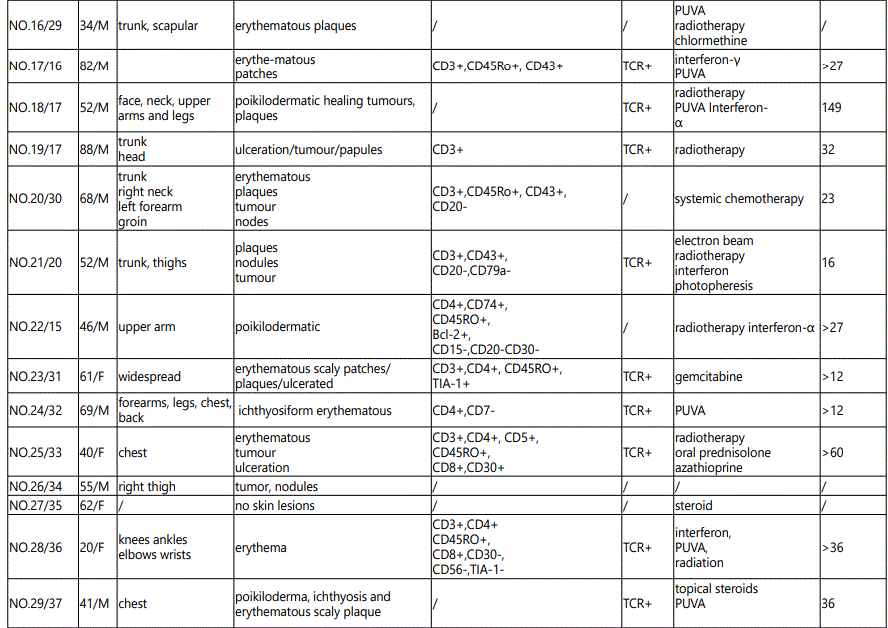
Since the first case of GMF in 1970, at least 41 cases have been reported in the literature. We summarized the clinicopathologic characteristics, prognosis, and therapeutic responses of GMF (Table 1).
GMF usually presented as non-specific skin lesion such as papule, macule, plaque, desquamation asymptomatic nodule or tumor. Acquired ichthyosis is supposed to be one of the cutaneous manifestations of a variety of malignancies [2]. In recent years, certain GMF cases with an ichthyosi form eruptions have also been reported [3].
Histopathological characteristics of GMF showed the basis of the typical mycosis granuloma (MF) with different quantities of granulomas, with tumor cells and epithelioid cells in filtrating, accompanied by elastic fiber atrophy. Granulomas can be observed in a variety of cutaneous T cell lymphomas (CTCLs), including sezary syndrome, primary cutaneous small/medium T cell lymphoma, primary cutaneous anaplastic large cell lymphoma, adult t-cell lymphoma, and primary cutaneous B-cell lymphomas [4-7]. However, GMF is the most common form of primary cutaneous malignant lymphoma showing granulomatous features [8]. About 2% of CTCLs cases express granuloma formation [9]. Characteristically, multinucleated giant cells showed emperipolesis of lymphocytesor containing phagocytized elastic fibers. Lymphocytic epidermotropism is almost always common. Immunopheno typing of the majority of GMF casesmanifest positive for CD3, CD4, CD5, CD45RO [10], while mostly negative for CD8, CD30 [11] verses extremely rare CD8-positive in GMF [12]. The analysis of T-cell clonality (TCR-PCR) is helpful for distinguishing granulomatous inflammatory reactions from granulomatous CTCLs, such as GMF.
GMF is easy to be misdiagnosed as inflammatory granuloma or infectious granulomas, such as granuloma annular (GA), sarcoidosis, and so on [13]. Histopathology, immunohistology and TCR gene analysis are helpful in differential diagnosis. A case of GMF presenting as sarcoidosis was reported in 1996 [14]. GMF and granulomatous slack skin (GSS) are both a lymphoma of T-cells, with similar histopathological and immune phenotype. GSS must be differentiated from GMF with the presence of marked epidermotropism, an extensive dermal infiltrate of granulomas, distinct elastophagocytosis and almost completely disappeared elastic fibers [8, 14-15]. However, it is still difficult to distinguish between the two diseases byhistopathology, while the clinical presentation of GSS is very characteristic, with bulky pendulous axillar and inguinal skin folds [8]. Some scholars hold that GMF is the early stages of GSS and both of them belong to a spectrum of the same entity [16]. Various therapies have been applied to treat GMF including PUVA, glucocorticoid, interferonalpha, retinoids, systemic chemotherapy, radiationand chemotherapy [17-18]. In the current case, we treat the patient with oral prednisoneandisotretinoin. The masses were mainly disappeared after one month of treatment. To consolidate the efficacy, isotretinoin had been continuously administrated for the next 6 months, while prednisone was tapered off after the first month treatment. In May 2013 (2 months after the regimen), a few local skin ulcers appeared on the chest and right lower limb (Fig.5).
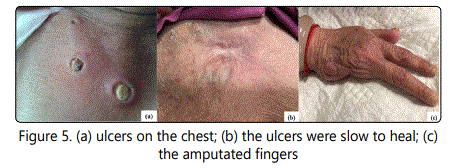
After a routine surgical treatment and hormone treatment, the ulcers slowly healed in one month. At present the patient was on prednisone (2.5 mg per day) as maintenance therapy. Patient has been free of tumorous symptoms for more than three years. Prednisone and isotretinoin regimen worked well in this case and avoid spectrum of side effects caused by systemic chemotherapy, radiation and chemotherapy.
The prognosis and clinical significance of granulomatous reaction in MF is not yet clear. Traditionally, granulomatous are an immune mechanism of self-protection, which is helpful for tumor regression and disease remission, and an indicator of favorable prognosis [7, 19]. However, many scholars consider that some patients of CTCL with granulomatous reaction express speedy development, high invasive, poor prognosis, may indicate disease is in progress to those with a poor outcome [11, 20]. Previous study reported that the 5-year-survival of GMF was 66%, which was worse than classic MF [9]. Nearly 40% of GMF patients died for their disease, and 50% of them died within 5 years of the onset of rashes [12]. In our case, during 52 months follow-up, the skin lesion progressed slowly with no other systemic involvement (Fig.5).
In this case, patient presented with non-specific skin lesion, infiltrative dark erythematous plaques, desquamation, nodule and tumor. Combining clinical manifestations, histopathological examination with TCR gene rearrangement results, the diagnosis of GMF was confirmed. Before the appearance of the rash, there was a mass in her left pinkie without symptoms in September 2008and it then spread to her left ring finger. Histopathological recorded chronic granuloma formation, which might be the early manifestation of GMF. Unfortunately, immunohistochemical and other further examinations were not taken at that time. The presence of granulomatous infiltrate in the early stage of GMF may result in a misdiagnosis. Identification in early stage is critical to prompt treatment and better prognosis, let alone that this patient presented with as rarely described icthyosi form skin eruption in its early stage. It is worth noticing that clinical heterogenicity of GMF is very common. The pathogenesis, the relationship among them, influences on the progression and prognosis are worthwhile studying thoroughly. In the current case, we noted that merely small doses of oral prednisone also achieved good therapeutic effect. Therefore, whether small doses of oral prednisone would be a reliable long-term maintenance treatment therapy for GMF, or only a part of heterogenicity of GMF?
Conflict of interest
The authors confirm that there is no conflict of interest regarding this manuscript.
References