Research Article
Gut Contents and Feeding Pattern of Macrobrachium vollenhovenii (HERKLOTS, 1857) and Caridina africana (Kingsley, 1882) at Asejire Lake and Erin-Ijesa waterfalls Southwest Nigeria
1Department of Agricultural Science, Osun State College of Education, Ilesa, Osun State, Nigeria
2Department of Aquaculture of Fisheries Management, University of Agriculture Abeokuta, Ogun State, Nigeria
*Corresponding author: Funmilayo Bosede Oyekanmi, Department of Agricultural Science, Osun State College of Education, Nigeria, Email: fumboye17@yahoo.com
Received: May 29, 2017 Accepted: June 10, 2017 Published: June 16, 2017
Citation: Oyekanmi FB, Omoniyi IT, AkegbejoSamsons Y. Gut contents and feeding pattern of Macrobrachium vollenhovenii (HERKLOTS, 1857) and Caridina africana (Kingsley, 1882) at Asejire Lake and Erin-Ijesa waterfalls Southwest Nigeria. Madridge J Aquac Res Dev. 2017; 1(1): 18-23. doi: 10.18689/mjard-1000104
Copyright: © 2017 The Author(s). This work is licensed under a Creative Commons Attribution 4.0 International License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
The gut contents and feeding pattern of (Macrobrachium vollenhovenii and Caridina africana) at Asejire Lake and Erin-Ijesa waterfalls respectively were studied for two seasons (October 2007 and September 2009) with intent towards acculturation. Sampling of each site for prawn composition and abundance was done twice every month. Catches obtained from the two areas were isolated and transported in ice chest boxes to the laboratory for numerical and biomass analysis of the gut contents. Data collected were subjected to one-way analysis of variance (ANOVA) and means were separated using Duncan New Multiple Range Test. There was a strong correlation (p<0.05) between prawn abundance and food availability. The gut content analysis of M. vollenhovenii showed that juveniles were predominantly zooplanktivorous feeding on copepods and rotifers while adult prawns were omnivorous feeding more on animal food than plant dietary items. This observation showed that prawns change diet as they advance in age. This has positive implication on its culture potential. Stomach fullness index was high during the wet season which coincided with high number of prawns. C. africana fed predominantly on diatoms and other micro-phytoplankton. The availability of both species in freshwater bodies and its food and feeding habit revealed that the species can be cultured in earthen ponds like its temperate counterpart Macrobrachium rosenbergii and can utilize artificial feeds for optimal productivity.
Keywords: Gut contents; Biomass analysis; Macrobrachium vollenhovenii ; Diet composition.
Introduction
Prawns which are valued food organisms are currently under-produced in Nigeria. The production and supply for local consumption is declining. There still exists wide gap between supply and demand, at least to meet FAO requirement for protein consumption [10]. Prawns are Omnivorous, and under natural condition feed on aquatic products. The natural foods of the freshwater prawns are vegetable waste, plankton, dead fishes, insect larvae, small worms and microorganisms [1]. But when farmed commercially, formulated feedstuffs are also provided. Natural food plays an important part in feeding especially young prawns. Food is held by the maxillipeds and torn into small pieces before being pushed into the mouth by the mouth parts [2].
Bello – Olusoji et. al. (2006) reported that prawns are zooplankton feeders with rotifers and copepods constituting the highest percentage occurrence in their stomach content and that there is a change in feeding habit from planktonivorous in the larvae to carnivorous in adult stages [3]. They further stated that prawns can function as primary consumer, secondary consumer and detrivore in aquatic system and hence be classified as omnivorous.
It has been reported that development of viable prawns farming requires effective feeding strategy which can be obtained through the knowledge of their food and feeding patterns in their natural habitat [2]. Therefore, this study was carried out on the food and feeding pattern in juvenile and adult Macrobrachium vollenhovenii and Caridina africana obtained from Asejire Lake and Erin-Ijesa waterfalls respectively.
Materials and Methods
Asejire Lake and Erin-Ijesa Waterfalls are located in Osun State Nigeria. Asejire Lake is man-made, constructed on river Osun. It lies on latitude 7o 23ʼ North and longitude 4o 05ʼ West. The Lake is Y-shaped with two unequal arms of the Y as shown in Figure 1. The catchment area of the dam is 7,800km2 and the impounded area is 2,342 hectares. The dam has a pool elevation (water level) of 150m and maximum flood elevation of 152.4m. The Lake has gross storage of 7,403 million litres.

Figure 1: Map Showing Asejire Lake
Erin-Ijesa waterfalls is situated in Oriade Local Government Area of Osun State within latitude 70 30′ and 80 45′ North and longitude 40 31′ and 50 0′ East. The inhabitants named the falls “Olumirin” the waterfalls have seven layers and only few visitors can climb beyond the second layer. Prawns are found more abundantly in the first, third and fifth layers the breeze at the waterfalls is cool and refreshing with average temperature variation of less than 250c. The water flows among the rock and splashes down with great forces to the evergreen vegetation around. The whole scenery is fascinating and ideal for mountain hiking, tourism and recreation. The location is shown in figure 2.
Figure 2: Map Showing Asejire Lake
The prawns were collected twice a month for each of the study sites for two hydrological cycles; from October 2008 –
September 2009 using baited traps at Asejire Lake while Plankton and hand nets were used to collect C. africana at Erin-Ijesa waterfalls. The samples were randomly selected and iced in order to reduce post-mortem digestion to minimum before analysis in the laboratory.
In the laboratory, the prawns were counted, sorted and identified based on taxonomic keys prepared by Reed et al (1967) and Lowe-McConnell (1972) [19] [23]. The stomach of each prawn specimen was dissected and the entire gut was emptied into the petri dish. Stomachs containing food were size classified according to a table from Olatunde (1978) [21]. The content of each stomach was weighed after being placed on a pile of filter papers that soaked away most of the moisture content. The content of all stomach were examined immediately or preserved in 4% neutral formalin for a later analysis. Fraction of food content placed on glass slides were observed under varying magnification of the microscope (MI-
1100LED). The food items were identified and in some cases up to generic levels.
Frequency of occurrence method were used by counting the number of stomachs in which each food item occurred, this was recorded and expressed as percentage of all nonempty stomach. Numerical abundance method was done by counting the number of individuals of each food type/
category in each stomach. These were added to give total for each kind of food item in the total samples examined and expressed in percentage as described by Bagenal and Tesch, (1978) [5]. The stomach contents were also identified based on methods described by Hyslop (1980), Imevbore (1965), Egborge (1972, 1973), Jeje and Fernando (1986) [8] [9] [13] [15] [16]. Occasionally, some food items were observed crushed and others were at varying stages of digestion. Consequently, it was not possible to identify these at the species level. These were measured as unidentifiable food items. The periodicity of feeding for juvenile and adult prawns was investigated as well.
Results
Specimens of Macrobrachium vollenhovenii and Caridina. africana, examined during the period of investigation showed that the condition of all stomachs varied from 1/4 to 4/4 full. Only 10% of the specimens had empty stomach suggesting that food was readily available for the species, it was observed that number of empty stomachs varied seasonality. Higher percentage of empty stomachs was observed in January and February, March, April and also in October, November and December. The month of intensive feeding was May, June, July, August, and September. This is shown in Table 1 & Fig 1. Tables 2 and 3 shows the frequency of occurrence and abundance in diet composition in the stomach of M. vollenhovenii and C. africana respectively. The blue green algae consisted of nostoc, Coelosphacnium and Aphanizomenon. The Green Algae included Memgeotia, Spirogyra, Protococcus, Coscinodiscs and Chaetocerus. The Protozoa was made up of Volvox, Ceratium, Fontonia and Diatoms (Diatoma). Rotifers consisted of Filinia, Rotaria and Keratella. The Clatoocerans on the other hand include Daphnia, Amphipods and the Copepods were made up of Cyclops. Fish remains and unidentified benthic matters (Detritus) were included in their food.
The preference of adult and juvenile for dietary items was almost the same, although the dietary spectrum of the adult was broader than that of juvenile, which fed mainly on zooplankton sources.
The adult M. vollenhovenii fed more on other dietary items such as protozoa, green algae, amphipods and fish remains that were not prevalent in the diet of juveniles. However, the food preference of adult and juvenile in C, africana did not show a marked pattern but their feeding range is relatively distributed among the dietary items.
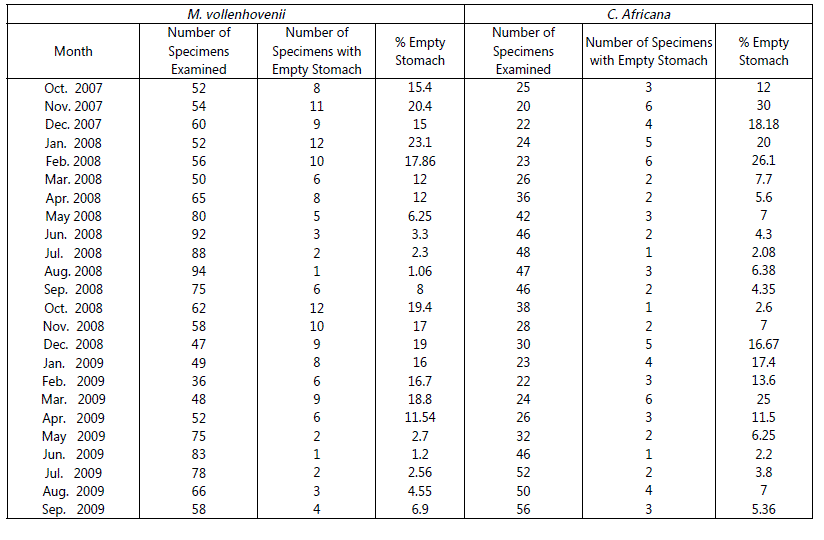
Table 1: Monthly changes in the number of empty stomach of Macrobrachium vollenhovenii and Caridina africana
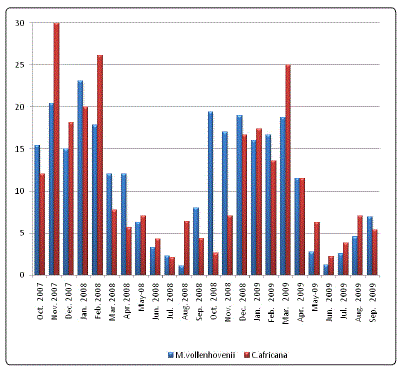
Figure 3: Monthly changes in the percentage of empty stomach of Macrobrachium vollenhovenii and Caridina africana
Discusion
Abundance of gut content composition increases during the rainy seasons as a result of nutrient upwelling caused by the rain. The gut content of the juveniles showed that they are zooplankton feeders with rotifers (19.3%) and copepods (18.5%) constituting the highest percentage of occurrence in the diet.
The food items in the stomach of adult M. vollenhovenii
suggest that they are euryphagous (i.e. feeding on a wide range of organisms). It was also observed that M. vollenhovenii
could be classified as an omnivorous feeder as the diet covers a wide spectrum of food ranging from various types of plankton to invertebrates and plants. Bello-Olusoji et al. (2006) described palaemonid prawns as non-selective feeders [3]. Fagade (1983) and Omoniyi & Bakare (1998) observed same with feeding habit of tilapia species [11] [22].
The prawn also exhibits an overlapping in food and feeding habits in order to avoid inter- and intra-specific competition for available food. C.africana fed on macrophages, diatoms and other unidentifiable benthic micro organisms. They exhibited nearly similar feeding pattern with M vollenhovenii
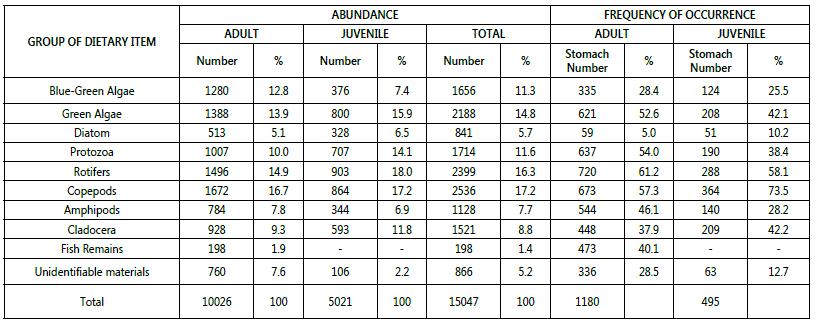
Table 2: Frequency of occurrence and abundance in diet composition in the stomach of Macrobrachium vollenhovennii in Asejire Lake.

Table 3: Frequency of occurrence and abundance in diet composition in the stomach of Caridina africana in Erin-Ijesa Waterfalls.
Monthly variations in feeding habits of the prawns showed an increase in the stomach fullness during the rainy season and decreases in the dry season. The proportion of empty stomachs in the juveniles was higher during the dry season. This may reflect a steady dwindling of food resources in a habitat that is continually decreasing in volume with the onset of the dry season. However, the proportion of empty stomachs were low, the percentage may reflect that the periods feeding was short followed by longer periods of rapid digestion. This may suggest that the two prawns feed intermittently and or have a high rate of digestion. Other reasons for this may be; during handling and transportation some food items may be quickly or partly digested thus making identification difficult. Moreover with high rates of digestion, some food practices may be difficult to observe or identify if the prawn has been caught in the traps for several hours, while digestion during this time can also reduce stomach fullness. These may account for the reduced stomach fullness found among the evening collections, because the prawns might have been trapped for longer period of time. Another possibility is that the fraction of empty stomachs observed may be biased upward owing to sampling with passive gear (baited nonreturned traps) as opposed to active gear (towing plankton net) because hungry prawns are more likely to take bait from a baited trap than satiated animals. According to Bello-Olusoji et al (2006) most aquatic animalʼs species appear to be opportunistic feeders consuming a large diversity of prey [3].
Periodicity variation shows that more prawns were caught in the night than the morning collection. Prawns collected during the wet season especially at the peak of the wet season (August-
October) had fuller gut contents than collections made during the dry season (February-April).more food availability occur during the rainy season than dry season. This perhaps suggests increase the prawn population during raining season. Moreover morning collections during the dry season had fuller gut content than collections made in the evening collection. This is an indication that, the animals are nocturnal and night feeders when they are more active searching for foods unlike in the day when they hide. In order words, the stomachs fill up earlier and remain full over a longer period. Thus increase in food abundance during the raining season, justified fuller stomach contents than dry season catches. Another possible reason why there was higher amount of food in their guts was due to the heavy and incessant rains, there is a lot of nutrients upwelling and a lot of washing of the substratum to the water bodies by run off; all resulting in increased availability of food for the prawns. The visibility remains poor and turbidity high for most of the day and this could induce feeding in prawns if there was food. This over-night periodicity tests show that prawns stomach is cleared each day when they buried themselves in the bottom sediments, or hide during the day when they are less catchable and came out to feed at night when most of them were caught.
Some of the variability in the dietary composition of M. vollenhovenii could be explained on the basis of the change in water level since the planktons have their preferred water condition. Seasonal variation occurs in the composition of the diet of the M.vollenhovenii because availability of food organisms is often cyclic due to factors of their life histories, climate, or other environmental conditions. Seasonal variation in the feeding habits of fish resulting from environmental changes has been reported by Moriarty and Moriarty (1973), Ikusemiju (1975), Tudorancea et al. (1988) [14] [24] [19]. The diet and feeding intensity can vary even during the diurnal cycle [7] [17]. During the rainy season, there is a wide variety and abundance of food available due to high nutrient brought by the run-off from land promoting plant growth and increasing invertebrate productivity [20]. This is reflected in the range of food items found in M. vollenhovenii captured during the rainy season (i.e June to September) supplying the Lake. As the dry season sets in the Lake becomes shallow and the abundance and variety of food decrease. This agrees with the findings of Hyslop (1986) [12]. However there is no marked pattern of seasonal variation in adult prawn, this may reflect that they do not have a preferred food items but feed on available diet irrespective of the season. It implies that the juvenile are stricter in their preference for food than the adult.
From the report of Wassenberg and Hill, (1987), it was also seen clearly that juvenile do not feed on fish and other bigger animals, though amphipods and polychaetes parts were found in the gut content of advance post-larvae, an indication that they are changing diets [25]. The shift from diets of animal origin as they grow older is also well known among penaeids. In the early adult, the proportion of animal organism in the diet showed an increase, though the early adult prawns were still zooplanktivorus, but the animal contents increases, until it became more pronounced in the mature adults. This shows a change in feeding habits, with all kinds of detritus, benthic organisms, phytoplankton, zooplankton and animal matter found in the gut. This view was supported by Costa and Wanninayake (1986) who had a similar observation in M. rosenbergii [6]. This observation can be inferred that prawn can function as a primary consumer, secondary consumer and detritus feeder in the aquatic system, and hence could be classified as ominivorous. However, Bello-Olusoji et al. (1995) reported that stomach contents of the juvenile prawns (5.00 –
7.80cm in length) comprised remains of benthic animals and fish as well as detritus which is an indication that they are benthic or detritus feeders/scavengers [4]. They also remarked that polychaete, small crustacean and fish remains constituted a proportion of not less than 40.85% of its food.
Costa and Wanninayake (1986) observed also that adult prawns have preference for more animal food items than plant matters [6]. Tripathi (1990) further established that M. rosenbergii is an omnivorous animal.
In addition to confirming the main dietary components, this study has shown four important features:
a) The periodicity of feeding of the apparent effect of season on feeding pattern,
b) The changes in the diet as the prawn get larger.
c) The apparent lack of habitat difference in diet from the two water bodies considered in this research
d) The similarities of the diet and feeding patterns of the prawns irrespective of species, age and the environment.
Conclusion and Recommendations
The results of the stomach content analyses showed that the M. vollenhovenii were omnivorous because they consume both plant and animal material as food. C.africana fed on macrophages, diatoms and other unidentifiable benthic microorganisms which are sometimes referred to as detritus. They exhibit almost similar feeding pattern with M vollenhovenii. Adult prawns of both species were non-selective and opportunistic feeders.
Conflict of Interest
The authors confirm that there is no conflict of interest regarding this manuscript.
References
- Arrignon JCV, Hunner JV, Laurent PJ, Griessiner JM, Lacroix D, et.al. Warm-Water Crustaceans. London.1994
- Brown L. Aquaculture for veterinarians. Fish husbandry and medicine pergamon press, Oxford New York, Seoul, Tokyo: 27-30.1993
- Bello-Olusoji OA, Bankole M, Sheu A, and Oyekanmi FB. Availability, Diet Composition and Feeding Behaivours of Some Commercially Important Paleamonidae Prawns in Fresn and Brackish Water of Nigeria. J. Biol. Sci. 2006; 6(1): 15-21.
- Bello-Olusoji AO, Balogun AM, Fagbenro OA, and Ugbaja N. Food and Feeding Studies of the African river prawn Macrobracium vollenhovenii, in Lavens, P., Jaspas, E. and Roelants, I. (eds). Larviʼ 1995 Fish and Shell Fish Larviculture Symposium. J. Europ. Aquac. Soc. 1995; 24: 425-427.
- Bagenal TB, Tesch FW. Age and Growth, In: T.B. Bagenal (ed.) Methods of Assessment of fish production in freshwaters. Oxford, Blackwell Scientific Publications. 44-45.1978
- Costa HH, Wanninayake TB. Food, feeding and fecundity of the giant freshwater prawn Macrobrachium rosenbergii from Natural Habitt in Sri Lanka, 555-558. In: The first Asian Fisheries Forum, Asian Fisheries Society. Manilla, Philippines. Eds. J. L. Maclean, L. B. Drion and Hosillo. Ministry of Agriculture, Manilla, Phillipines. 1986.
- Elliot OO. Diet changes in invertebrate drift and the food of trout, Salmon trutta. J. Fish. Biol. 1970; 2: 1-18.
- Egborge ABM. Checklist of zooplankton in River Oshun. Nig. J. Sci. 1972; 8: 51-62.
- Egborge ABM. A preliminary checklist of the phytoplankton of the Osun River Western Nigeria. Freshwater. Bio. 1973; 3: 569-572.
- F.A.O (2000). The state of world Fisheries and Aquaculture, Rome, Italy.
- Fagade SO. The biology of Chromidotilapia guentheri from a small Lake. Arch. Hydrobiol. 1983; 97: 60-72.
- Hyslop EJ. The growth and feeding habits of Clarias anguillaris during their first season in the flood plain pools of the Sokoto-Rima River Basin, Nigeria. J. Fish. Biol. 1986; 30(2): 183-192. doi: 10.1111/j.1095-8649.1987. tb05744.x
- Hyslop EJ. Stomach contents analysis – a review of methods and their application. J. Fish. Biol. 1980; 17(4): 411-429. doi: 10.1111/j.1095-8649.1980.tbo2775.x
- Ikusemiju K. Aspects of the ecology and history of the Scopon cottus aletiticus (gilbert) in Lake Washington. J. Fish. Biol. 1975; 7: 235-245.
- Imevbore AMA. A preliminary checklist of the plankton organisms of Eleiyele reservoir, Ibadan, Nigeria. J. W. Afri. Sci. Ass. 1965; 10: 56-60.
- Jeje CY, Fernando CH. A practical guide to the identification of Nigerian Zookplanton (cladocera, copepod and Rotifera). A publication of Kainji Lake Research Institute (KLRI), New Bussa.1986
- Keast A, Welsh L. Daily feeding periodicities, food uptake rate and dietary changes with hour of the day in some Lake fishes. J. Res. Fisheries. Bod. Can. 1968; 25(6): 1133-1144. doi: 10.1139/f68-099
- Lowel-McConnell RH. Freshwater fishes of the Volta and Kainji Lakes, Accra, Ghana. University Press. 1972
- Moriarty CM, Moriarty DSW. Quantitative estimation of the daily ingestion of phytoplankton by Tilapia nilotica and Haplochronis nigripinis in Lake George, Uganda. J. Zool. 1973; 171(1):15-23. doi: 10.1111/j.1469-7998.1973.tb07513.x
- Moss B. Ecology of freshwaters. Blackwell Scientific Publication, Oxford. 1980
- Olatunde AA. The Food and Feeding Habits of Eutropius niloticus (Ruppel), family Schilbeidae (Osteichthyes: Siluriformes) in Kainji Lake, Nigeria. Hydrobiologia, 1978; 57: 197–207.
- Omoniyi IT, Bakare O. Natural Diets and Length-Weight Relationship of Sarotherodon Galilaeus (L) in Ado-Ekiti Water Reservoir, Ekiti state of Nigeria, Global. J. Pure & Appl. Sci. 1998; 4(4): 343–347.
- Buchard RW, Hopson J, Jenness AJ, Yaro I. Fish and Fisheries of Northern Nigeria. Ministry of Agriculture, Zaria. 262p.1967
- Tudorancea CJ, Fermado CN, Paggy JC. Food and feeding ecology of Oreochromis niloticus (Linnaeus, 1758) jóvenes in Lake Awasa (Ethiopia). j. Arch. Hydrobiol. 1988; 79: 267-289.
- Wassenberg TJ, Hill BJ. Natural diet of the tiger prawns Penaeus esculentus and P semisulcatus. J. mar. Freshwater. Res. 1987; 38: 169-182. doi:10.1071/MF9870169


